��Ŀ����
�����й�ʵ���˵����ȷ����
A������K2Cr2O7��Һ�м��������������ƣ���Һ��Ϊ��ɫ��֤����Һ�д�����ƽ�⣺Cr2O72-+H2O 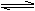 2CrO42-+2H+ 2CrO42-+2H+ |
| B��CH2��CH��CHO��ʹ���Ը��������Һ��ɫ��֤���÷�����һ������̼̼˫�� |
| C������μӵķ������ܼ���NaCl��AlCl3��Na2CO3��NaHSO4������ɫ��Һ |
| D��SO2ͨ��Ba(NO3)2��Һ�г��ְ�ɫ������֤��BaSO3������ˮ |
A
������ȷ�𰸣�A��Cr2O72-+H2O 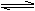 2CrO42-+2H+����NaOH��ƽ�������ƶ����ɳ�ɫ��Ϊ��ɫ��B��̼̼˫����ȩ������ʹ���Ը��������Һ��ɫ��C��Na2CO3��NaCl��AlCl3��NaHSO4���õ�����ֱ�������������ɫ���������ݡ����ݣ�D������ȷ���������ɵ���BaSO4,���������£�NO3�D�ܽ�SO2������SO42�D.
2CrO42-+2H+����NaOH��ƽ�������ƶ����ɳ�ɫ��Ϊ��ɫ��B��̼̼˫����ȩ������ʹ���Ը��������Һ��ɫ��C��Na2CO3��NaCl��AlCl3��NaHSO4���õ�����ֱ�������������ɫ���������ݡ����ݣ�D������ȷ���������ɵ���BaSO4,���������£�NO3�D�ܽ�SO2������SO42�D.

��ϰ��ϵ�д�
�����Ŀ
�����ʽṹ�����ʡ�
�����������ʵ���Ҫ���������ʽṹ����ش��������⡣
��1����2�֣�CH3������CH3��CH3��������Ҫ���л���Ӧ�м��壬�й����ǵ�˵����ȷ����
| A�����Ǿ��ɼ���ȥ��һ����ԭ������ |
| B�����ǵĿռ乹����ͬ��̼ԭ�Ӿ����Բ�ȡsp2�ӻ� |
| C��CH3����NH3��H3O+��Ϊ�ȵ����壬���ι��;�Ϊ������ |
| D��CH3���е�̼ԭ�Ӳ�ȡsp3�ӻ�������ԭ�Ӿ����� |
 ����q���йأ�һ����ż���أ��̣������������ӵ�ż���ض���Ϊż������ż����һ�˵�ɵ����ij˻������̣�d��q���Իش��������⣺
����q���йأ�һ����ż���أ��̣������������ӵ�ż���ض���Ϊż������ż����һ�˵�ɵ����ij˻������̣�d��q���Իش��������⣺��HCl��CS2��H2S��SO2���ַ����Ц̣�0���� ��
��ʵ���ã���PF3
 ��1.03����BCl3��0���ɴ˿�֪��PF3������ ���ͣ�BCl3������ ���͡�
��1.03����BCl3��0���ɴ˿�֪��PF3������ ���ͣ�BCl3������ ���͡���3����2�֣�
 �����ʵĴ����о����������������Ӻ�δ�ɶԵ���Խ�࣬�����Խ�ż�¼����Խ�á�������������V2O5��CrO2�У��ʺ���¼�����ŷ�ԭ�ϵ���__________________��
�����ʵĴ����о����������������Ӻ�δ�ɶԵ���Խ�࣬�����Խ�ż�¼����Խ�á�������������V2O5��CrO2�У��ʺ���¼�����ŷ�ԭ�ϵ���__________________����4����3�֣����ù��������֤ʵ
 ���ڽྻ���������ںϳɰ�
���ڽྻ���������ںϳɰ���Ӧ�Ĵ������ı����ϴ��ڵ�ԭ�ӣ���ͼΪ��ԭ�������ľ����ϵĵ��㸽�žֲ�ʾ��ͼ��ͼ��С��ɫ�������ԭ�ӣ���ɫ�������ԭ�ӣ������ڵ��㾧����N/Feԭ����֮��Ϊ________________��

��5����2�֣���������Ľṹ���õȾ�Բ����ܶѻ����������ڵȾ�Բ������ܶѻ��ĸ�����ʽ�У��������ܶѻ����������ܶѻ���Ϊ��Ҫ����ָ����ͼ���ĸ�Ϊ�������ܶѻ� ���A����B����

ͼA ͼB


 �����ʵĴ����о����������������Ӻ�δ�ɶԵ���Խ�࣬�����Խ�ż�¼����Խ�á�������������V2O5��CrO2�У��ʺ���¼�����ŷ�ԭ�ϵ���__________________��
�����ʵĴ����о����������������Ӻ�δ�ɶԵ���Խ�࣬�����Խ�ż�¼����Խ�á�������������V2O5��CrO2�У��ʺ���¼�����ŷ�ԭ�ϵ���__________________��


