��Ŀ����
10�� ij��Һ�п��ܺ���H+��Na+��NH4+��Fe3+��Al3+��SO42-��CO32-�����ӣ��������Һ�м���һ�����ʵ���Ũ�ȵ�NaOH��Һʱ���������ɳ��������ʵ�����NaOH��Һ������仯��ͼ����ͼ��ʾ������˵������ȷ���ǣ�������
ij��Һ�п��ܺ���H+��Na+��NH4+��Fe3+��Al3+��SO42-��CO32-�����ӣ��������Һ�м���һ�����ʵ���Ũ�ȵ�NaOH��Һʱ���������ɳ��������ʵ�����NaOH��Һ������仯��ͼ����ͼ��ʾ������˵������ȷ���ǣ�������| A�� | ԭ��Һ��һ�����е���������H+��NH4+��Fe3+��Al3+ | |
| B�� | ԭ��Һ��һ������SO42- | |
| C�� | ԭ��Һ�к��е�Fe3+��Al3+�����ʵ���֮��Ϊ1��1 | |
| D�� | ��Ӧ����γɵ���Һ������ΪNa2SO4 |
���� A���������ɳ��������ʵ�����NaOH��Һ������仯��ͼ����ȷ�����ڵ����ӣ�
B�����ݵ���غ���Aѡ���������ȷ�����ڵ����ӣ�
C�����������������Ƶ��������ɳ���������ȷ�����ӵ����ʵ����Ķ��٣�
D��������Һ������֮��ķ�Ӧ��ȷ�����е����ʣ�
��� �⣺A������ͼ���һ�Σ������������ϳ��֣���֪һ���������ӣ�����кͷ�Ӧ��һ�з�Ӧ�����ȷ�Ӧ�����ɳ����ں����ܽ�һ���֣�һ���������ӣ����ݵ����Σ����������Ʒ�Ӧ�����Ӳ�����������֤����笠����ӣ�������������ɳ�������6������������ƣ��������������ܽ�����һ������������ƣ������γ���������������������������ƣ��������������������һ���DZ��������������ģ�����һ���������������ӣ�һ������̼������Ӻ�þ���ӣ���ԭ��Һ�к��е���������H+��NH4+��Fe3+��Al3+����A��ȷ��
B������A���ƶϣ�����ͼ��ڶ��κ����ʣ���������������֪�����������ij���������һ������������ȵģ���һ�������������ӣ�һ��������þ���ӣ����Ժ��е���������H+��NH4+��Fe3+��Al3+��һ������̼������ӣ���Һ����Ҫ���������ӣ���һ����������SO42-������ȷ���������Ƿ���ڣ���B��ȷ��
C��������������ɳ�������6������������ƣ��������������ܽ�����1������������ƣ������γ�������������3������������ƣ�����3�������������һ���DZ��������������ģ�ԭ��Һ�к��е�Fe3+��Al3+�����ʵ���֮��Ϊ1��1����C��ȷ��
D������NaOH��Һ��������ʵ�ʲ��μӷ�Ӧ����Ӧ����Һ��������һ�����ڣ������Һ�к��е�����ΪNa2SO4��ƫ�����ƣ���D����
��ѡD��
���� ���⿼���˳������ӵ����ʼ����鷽������Ŀ�Ѷ��еȣ�ע�����ճ������ӵ����ʼ����鷽������ȷ��Ԫ�ؼ��仯��������Ϊ���ؼ�����������ȷ�жϴ��������Ӷ�������þ����Ϊ�״��㣬��Ҫ��������ͼ�����߱仯�����ݣ�

 �������±������ϣ��ش��������⣺
�������±������ϣ��ش��������⣺�������������Ŀ���ʴ����Խ��Խ�ܵ����ǵ��i��������Ҫ����������������ͭ��̼��Ԫ������ɵĺϽ�
���ࣨGe���ǵ��͵İ뵼��Ԫ�أ��ڵ��ӡ����ϵ�����Ӧ�ù㷺��
��K2Cr2O7�����ڼ��˾���Ƿ�ƺ��ʻ��
Cr2O72-����ɫ��+CH3CH2OH��Cr3+����ɫ��+CH3COOH ��δ��ƽ��
��1����Ԫ�ػ�̬ԭ�ӵĵ����Ų�ʽΪ1s22s22p63s23p63d84s2��[Ar]3d84s2��
��2��CH3COOH����������Ԫ�صĵ縺���ɴ�С��˳��ΪO��C��H��̼ԭ�ӵĹ���ӻ�����Ϊsp3��sp2�������Ҽ���м�����Ŀ֮��Ϊ7��1��
��3���Ƚ�������±������۵�ͷе㣬������仯���ɼ�ԭ��GeCl4��GeBr4��GeI4���ۡ��е��������ߣ�ԭ���Ƿ��ӽṹ���ƣ����������������Ӽ������������ǿ��
| GeCl4 | GeBr4 | GeI4 | |
| �۵�/�� | -49.5 | 26 | 146 |
| �е�/�� | 83.1 | 186 | Լ400 |
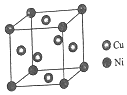
�پ�����ͭԭ������ԭ�ӵ�������Ϊ3��1��
�����Ͻ���ܶ�Ϊdg/cm3����������a=$\root{3}{\frac{251}{d{N}_{A}}}$��107nm��
| A�� | pH=7����Һ��Fe3+��NH4+��Cl-��NO3- | |
| B�� | ˮ�������c��H+��=$\sqrt{{K}_{w}}$����Һ�У�Na+��SO42-��CO32-��K+ | |
| C�� | ��ˮ�������c��OH-��=1.0��10-2 mol•L-1����Һ��Cl-��CH3COO-��K+��[Al��OH��4]- | |
| D�� | $\frac{[O{H}^{-}]}{[{H}^{+}]}$=1012����Һ�У�NH4+��Al3+��NO3-��Cl- |

��1�������Ԫ�ؽ����ʵĴ�ʩ����дһ�������Ȼ������ֽ���ȣ�
��2������1�У�������ת��Ϊ���κ������Σ���д��������Ӧ�Ļ�ѧ����ʽ2FeS2+15MnO2+14H2SO4=Fe2��SO4��3+15MnSO4+14H2O��
��3���ӹ�ҵ�����ɱ����ǣ������A�����A��
A��CaCO3 B��NaOH C����ˮ D��Ca��OH��2
��4������Һ2����Ҫ�ɷ�Ag��CN��2-��д������2�з�����Ӧ�����ӷ���ʽ4Ag+O2+8CN-+2H2O=4 Ag��CN��2-+4OH-���������Һ�У�����ѭ�����õ�����NaCN����CN-�����ѧʽ����
��5����ͼ2��֪����Һ�̱ȣ�ˮ����ϵ������ȣ�����5ʱ�������ʷ������͵�ԭ��NaCN Ũ�Ƚ��ͣ���ȡ���ʵͣ�
��6�����̿��У�������������״��ɢ��MnO2�����У����ƻ�ԭ�нṹ��������������̵Ľ�������������ȡ��������Ҫ��ijС��ͨ��ʵ�飬�о�������������������������ռ���ذٷֱȣ����̽����ʵ�Ӱ�죬�����ʾ��
| ����% | 31.52 | 36.10 | 40.67 | 45.25 |
| �̽����� | 77.42 | 89.49 | 91.59 | 97.95 |
| ������% | 12.5 | 15 | 17.5 | 20 |
| �̽����� | 88.79 | 97.95 | 99.18 | 99.46 |
| A | ����45.25% | B | ����31.52% | C | ����45.25% | D | ����36.10% |
| ������20% | ������15% | ������15% | ������20% |
| A�� | CH4+C12$\stackrel{��}{��}$ CH3Cl+HCl | |
| B�� | CH2=CH2+H2O��CH3-CH2OH | |
| C�� |  +Br2 $\stackrel{FeBr_{3}}{��}$ +Br2 $\stackrel{FeBr_{3}}{��}$  +HBr +HBr | |
| D�� | 2 CH3CH2OH+O2$��_{��}^{Cu}$ 2 CH3CHO+2 H2O |
��ҵ��ͨ���ýӴ��������ᣬ��Ҫԭ����������Ϳ������Ӵ�����������������̴��¿� ��Ϊ�����Σ������������ȡ�;�������������ת��Ϊ��������������������պ���������ɣ�Ϊ�˷�ֹ������Ⱦ����β�������ۺ����ã����᳧���ð�ˮ����β����SO2��SO3�����壬��������Һ�м���Ũ���ᣬ����ȡ��Ũ�ȵ�SO2����NH4��2SO4��NH4HSO4�� �壮Ϊ�˲ⶨ�ⶨ������NH4��2 SO4��NH4HSO4�����������ɣ��ֳ�ȡ����Ʒ�ķݣ��ֱ� ������ͬŨ�ȵ�NaOH��Һ50.00mL��������120�����ң�ʹ����ȫ���ݳ�[��NH4��2 SO4�� NH4HSO4�ķֽ��¶Ⱦ�����200��]������й�ʵ���������£���״������
| ʵ�� | ��Ʒ������/g | NaOH��Һ�����/mL | ���������/L����״���� |
| 1 | 7.24 | 50.00 | 1.792 |
| 2 | 14.48 | 50.00 | 3.584 |
| 3 | 21.72 | 50.00 | 4.032 |
| 4 | 36.20 | 50.00 | 2.240 |
��2���Լ���û�����У�NH4��2SO4�� NH4HSO4�����ʵ���֮��Ϊ1��2��
��3��������NaOH��Һ�����ʵ���Ũ�ȣ�
