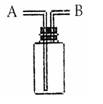
��Ŀ����
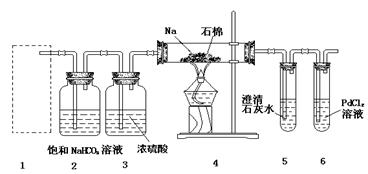
ij����С����MnO2��Ũ�����Ʊ�Cl2ʱ�����ø����չ�����SO2��NaOH��Һ����β���������մ�����
��1�������SO2��������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ�� ��2�֣�
��2����ӦCl2+Na2SO3+2NaOH===2NaCl+Na2SO4+H2O��ÿת��2.5mol�ĵ�����μӷ�Ӧ�Ļ�ԭ�������ʵ���Ϊ mol����2�֣�
��3������β��һ��ʱ�������Һ��ǿ���ԣ��п϶�����Cl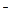 ��OH
��OH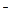 ��SO
��SO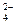 �������ʵ�飬̽��������Һ�п��ܴ��ڵ����������ӣ������ǿ�����CO2��Ӱ�죩��
�������ʵ�飬̽��������Һ�п��ܴ��ڵ����������ӣ������ǿ�����CO2��Ӱ�죩��
������������� ��
����1��ֻ����SO32-�� ����2��ֻ����ClO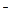
����3���Ȳ�����SO32-Ҳ������ClO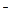 ��
��
����4�� ����2�֣�
�����ʵ�鷽������ʵ�顣���ڴ����д��ʵ�鲽���Լ�Ԥ������ͽ���
��ѡʵ���Լ���3moL L-1H2SO4��0.01mol
L-1H2SO4��0.01mol L-1KMnO4����ɫʯ����Һ����ÿ��2�֣�
L-1KMnO4����ɫʯ����Һ����ÿ��2�֣�
��1�������SO2��������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ�� ��2�֣�
��2����ӦCl2+Na2SO3+2NaOH===2NaCl+Na2SO4+H2O��ÿת��2.5mol�ĵ�����μӷ�Ӧ�Ļ�ԭ�������ʵ���Ϊ mol����2�֣�
��3������β��һ��ʱ�������Һ��ǿ���ԣ��п϶�����Cl
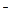 ��OH
��OH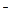 ��SO
��SO �������ʵ�飬̽��������Һ�п��ܴ��ڵ����������ӣ������ǿ�����CO2��Ӱ�죩��
�������ʵ�飬̽��������Һ�п��ܴ��ڵ����������ӣ������ǿ�����CO2��Ӱ�죩��������������� ��
����1��ֻ����SO32-�� ����2��ֻ����ClO
����3���Ȳ�����SO32-Ҳ������ClO
 ��
������4�� ����2�֣�
�����ʵ�鷽������ʵ�顣���ڴ����д��ʵ�鲽���Լ�Ԥ������ͽ���
��ѡʵ���Լ���3moL
 L-1H2SO4��0.01mol
L-1H2SO4��0.01mol L-1KMnO4����ɫʯ����Һ����ÿ��2�֣�
L-1KMnO4����ɫʯ����Һ����ÿ��2�֣�| ʵ�鲽�� | Ԥ������ͽ��� |
����1��ȡ��������Һ���Թ��У��μ�3 moL L-1 H2SO4����Һ�����ԣ�Ȼ��������Һ������A��B�Թ��У� L-1 H2SO4����Һ�����ԣ�Ȼ��������Һ������A��B�Թ��У� | |
| ����2�� | |
| ����3�� | |
(1) SO2+NaOH=NaHSO3(2��); (2)1.25mol(2��)
(3) ��SO32-��ClO-������(2��)
��(ÿ��2��)
(3) ��SO32-��ClO-������(2��)
��(ÿ��2��)
| ʵ�鲽�� | Ԥ������ͽ��� |
����1��ȡ��������Һ���Թ��У��μ�3 moL L-1 H2SO4����Һ�����ԣ�Ȼ��������Һ������A��B�Թ��� L-1 H2SO4����Һ�����ԣ�Ȼ��������Һ������A��B�Թ��� | |
| ����2����A�Թ��еμ���ɫʯ����Һ | ���ȱ�����ɫ��֤����ClO-�������� |
����3����B�Թ��еμ�0.01mol L-1KMnO4��Һ L-1KMnO4��Һ | ���Ϻ�ɫ��ȥ��֤����SO32-�������� |
��

��ϰ��ϵ�д�
 ֥�鿪���γ�������ϵ�д�
֥�鿪���γ�������ϵ�д�
�����Ŀ
|
����ʵ���������������ȷ���ǣ� �� 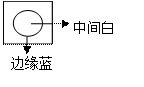 �ٽ�NaNO3��KCl�Ļ��Һ���Ȳ�Ũ�����о������������ȹ���ʱ���ɷ����NaCl���� �ں����е�Ԫ�صķ��뼰����ʱ����Ҫ���ҵĽ�ȡҺ�м�������ϡ�������������Һ ��ֽ���������������Ӻ�ͭ����ʵ���У����������ֽ�����ɺ��ܽ������չ������ �ܾ���������������ˮ������������Na2CO3��Һ��FeSO4��Һ����ʹ������Һ���� ���ü��ȷ����Է��������غ͵ⵥ�ʵĻ�����Ϊ�ⵥ������������ ������һ�����ʵ���Ũ�ȵ���Һʱ������ȡҺ̬���ʵ���Ͳ��ˮϴ�ӣ�ϴ��Һ��������ƿ �߽�������ˮ�ε����۵⻯����ֽ�ϳ�����ͼ����˵����Ũ����ˮ�ܽ�I-������I2����Ũ����ˮ�ܽ�I2��һ�������ɵ�Ļ�����  �ཫ����CO2ͨ��Ca(ClO)2��Һ�ó�����Һ��˵��H2CO3�����Ա�HClO�� 
|
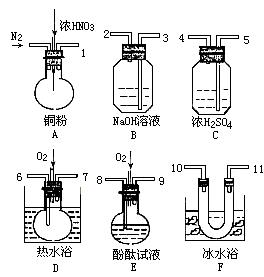
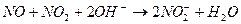 �� ����Һ���¶ȣ�NO2 21�棬 NO -152��
�� ����Һ���¶ȣ�NO2 21�棬 NO -152�� ����̼̼˫�������Ա�һ����ʹ ��ɫ��
����̼̼˫�������Ա�һ����ʹ ��ɫ�� ��̼ԭ��֮��Ľ� ��6��̼ԭ�Ӻ�6����ԭ�Ӷ���ͬһ �ϡ�
��̼ԭ��֮��Ľ� ��6��̼ԭ�Ӻ�6����ԭ�Ӷ���ͬһ �ϡ� һ���̼̼����˫����Ļ�״�ṹ��
һ���̼̼����˫����Ļ�״�ṹ��