��Ŀ����
�±���25��ʱijЩ�ε��ܶȻ�����������ĵ���ƽ�ⳣ��������˵����ȷ����
|
��ѧʽ |
AgCl |
Ag2CrO4 |
CH3COOH |
HClO |
H2CO3 |
|
Ks�� Ka |
Ksp=1.8��10-10 |
Ksp=2��10-12 |
Ka=1.8��10-5 |
Ka=3.0��10-8 |
Ka1=4.1��10-7 Ka2=5.6��10-11 |
A����ͬŨ��CH3COONa��NaClO�Ļ��Һ�У�������Ũ�ȵĴ�С��ϵ��c��Na+��>c��ClO-��>c��CH3COO-��>c��OH-��>c��H+��
B����0.1mol��L-1CH3COOH��Һ�еμ�NaOH��Һ��c(CH3COOH)��c(CH3COO-)=5 ��9����ʱ��ҺpH=5
C��̼������Һ�еμ�������ˮ�����ӷ���ʽΪCO32-+Cl2=HCO-3+Cl-+HClO
D����Ũ�Ⱦ�Ϊ1��10-3mol��L-1��KCl��K2CrO4���Һ�еμ�1��10-3mol��L-1��AgNO3��Һ��CrO42-���γɳ���
B
������������ĵ��볣�����ڴ�����ģ����Դ������Ƶ�ˮ��̶ȴ��ڴ����Ƶģ�A����ȷ��Ӧ����c��Na+��>c��CH3COO-��>c��ClO-��>c��OH-��>c��H+����B����������Ũ��Ϊ5x�����������ӵ�Ũ��Ϊ9x�����ݵ��볣������ʽ��֪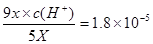 �����������Ũ����10��5mol/L������pH��5��B��ȷ��C��̼���ƹ�������������ĵ��볣������̼��ĵڶ������룬���Դ�������Ժ�̼���Ʒ�Ӧ���ɴ������ƺ�̼�����ƣ�C����ȷ�������ܶȻ�������֪ʹ�Ȼ���������������Ũ����1.8��10-7mol��L-1����ʹ������������������Ũ�ȴ���1.8��10-7mol��L-1������Ȼ����ȳ���������ѡ��D����ȷ����ѡB��
�����������Ũ����10��5mol/L������pH��5��B��ȷ��C��̼���ƹ�������������ĵ��볣������̼��ĵڶ������룬���Դ�������Ժ�̼���Ʒ�Ӧ���ɴ������ƺ�̼�����ƣ�C����ȷ�������ܶȻ�������֪ʹ�Ȼ���������������Ũ����1.8��10-7mol��L-1����ʹ������������������Ũ�ȴ���1.8��10-7mol��L-1������Ȼ����ȳ���������ѡ��D����ȷ����ѡB��

��2012?�㽭ģ�⣩�±���25��CʱijЩ�ε�Ũ�Ȼ�����������ĵ���ƽ�ⳣ��������˵����ȷ���ǣ�������
|
�±���25��ʱijЩ�ε��ܶȻ�����������ĵ���ƽ�ⳣ��������˵����ȷ����
| ��ѧʽ | AgCl | Ag2CrO4 | CH3COOH | HClO | H2CO3 |
| Ks�� Ka | Ksp=1.8��10-10 | Ksp=2��10-12 | Ka=1.8��10-5 | Ka=3.0��10-8 | Ka1=4.1��10-7 Ka2=5.6��10-11 |
A����ͬŨ��CH3COONa��NaClO�Ļ��Һ�У�������Ũ�ȵĴ�С��ϵ��c��Na+��>c��ClO-��>c��CH3COO-��>c��OH-��>c��H+��
B����0.1mol��L-1CH3COOH��Һ�еμ�NaOH��Һ��c(CH3COOH)��c(CH3COO-)=5 ��9����ʱ��ҺpH=5
C��̼������Һ�еμ�������ˮ�����ӷ���ʽΪCO32-+Cl2=HCO-3+Cl-+HClO
D����Ũ�Ⱦ�Ϊ1��10-3mol��L-1��KCl��K2CrO4���Һ�еμ�1��10-3mol��L-1��AgNO3��Һ��CrO42-���γɳ���
�±���25��CʱijЩ�ε�Ũ�Ȼ�����������ĵ���ƽ�ⳣ��������˵����ȷ���ǣ� ��
|
��ѧʽ |
AgCl |
Ag2CrO4 |
CH3COOH |
HClO |
H2CO3 |
|
KSP��Ka |
KSP=1.8��10-10 |
KSP=2.0��10-12 |
Ka=1.8��10-5 |
Ka=3.0��10-8 |
Ka1=4.1��10-7 Ka2=5.6��10-11 |
A����ͬŨ��CH3COONa��NaClO�Ļ��Һ�У�������Ũ�ȵĴ�С��ϵ��
c��Na+��>c��ClO-��>c��CH3COO-��>c��OH-��>c��H+��
B��̼������Һ�еμ�������ˮ�����ӷ���ʽΪCO32-+Cl2=HCO3-+Cl-+HClO
C����0.1mol��L-1CH3COOH��Һ�еμ�NaOH��Һ��c��CH3COOH����c��CH3COO-��=5��9����ʱ��ҺpH=5
D����Ũ�Ⱦ�Ϊ1��10-3mol��L-1��KCl��K2CrO4���Һ�еμ�1��10-3mol��L-1��AgNO3��Һ��CrO42-���γɳ���
�±���25��ʱijЩ�ε��ܶȻ�����������ĵ���ƽ�ⳣ��������˵����ȷ����
| ��ѧʽ | AgCl | Ag2CrO4 | CH3COOH | HClO | H2CO3 |
| Ks�� Ka | Ksp=1.8��10-10 | Ksp=2��10-12 | Ka=1.8��10-5 | Ka=3.0��10-8 | Ka1=4.1��10-7 Ka2=5.6��10-11 |
B����0.1mol��L-1CH3COOH��Һ�еμ�NaOH��Һ��c(CH3COOH)��c(CH3COO-)="5" ��9����ʱ��ҺpH=5
C��̼������Һ�еμ�������ˮ�����ӷ���ʽΪCO32-+Cl2=HCO-3+Cl-+HClO
D����Ũ�Ⱦ�Ϊ1��10-3mol��L-1��KCl��K2CrO4���Һ�еμ�1��10-3mol��L-1��AgNO3��Һ��CrO42-���γɳ���