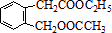��Ŀ����
ͼ��A��B��C��D��E��F��G��Ϊ�л������B������ֻ��һ����״�ṹ������ͼʾ�ش����⣺
��1��B�ķ���ʽ��______��A�Ľṹ��ʽ��______����Ӧ�ٵķ�Ӧ������______��
��2����Ӧ�۵Ļ�ѧ����ʽ��______��
��3������G���ʵ��йػ�ѧ����ʽΪ______��д1�����ɣ�
��4����������3��������B��ͬ���칹�����Ŀ��______��
�ٺ����ڶ�ȡ�������ṹ
����B����ͬ������
�۲���FeCl3��Һ������ɫ��Ӧ
д����������һ��ͬ���칹��Ľṹ��ʽ______��
��1��B�ķ���ʽ��______��A�Ľṹ��ʽ��______����Ӧ�ٵķ�Ӧ������______��
��2����Ӧ�۵Ļ�ѧ����ʽ��______��
��3������G���ʵ��йػ�ѧ����ʽΪ______��д1�����ɣ�
��4����������3��������B��ͬ���칹�����Ŀ��______��
�ٺ����ڶ�ȡ�������ṹ
����B����ͬ������
�۲���FeCl3��Һ������ɫ��Ӧ
д����������һ��ͬ���칹��Ľṹ��ʽ______��

��E�Ľṹ������֪��BΪ

�����F�ķ���ʽC4H8O2��C��Dת��ΪF�ķ�Ӧ����������֪CΪ���ᣬDΪ�Ҵ���GΪ��ȩ��FΪ������������BCD�Ľṹ�����A�ķ���ʽ��Aת��ΪBCD�ķ�Ӧ���������Ƴ�AΪ

��
��1��BΪ

������ʽΪC9H10O3��A�Ľṹ��ʽ��

����Ӧ��Ϊ����ˮ�ⷴӦ��
�ʴ�Ϊ��C9H10O3��

��ˮ�⣻
��2����Ӧ��Ϊ������Ҵ���Ũ���������·���������Ӧ����Ӧ�ķ���ʽΪCH3COOH+C2H5OH
CH3COOC2H5+H2O��
�ʴ�Ϊ��CH3COOH+C2H5OH
CH3COOC2H5+H2O��
��3��GΪ��ȩ������-CHO������������Һ����������Ӧ����Ӧ�ķ���ʽΪCH3CHO+2Ag��NH3��2OH
CH3COONH4+2Ag��+3NH3+H2O��
�ʴ�Ϊ��CH3CHO+2Ag��NH3��2OH
CH3COONH4+2Ag��+3NH3+H2O��
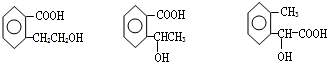
��4��BΪ

����Ӧ��ͬ���칹���У��ٺ����ڶ�ȡ�������ṹ��
����B����ͬ�����ţ�˵��ҳ����-COOH��-OH��
�۲���FeCl3��Һ������ɫ��Ӧ��˵���������ǻ�������ܵĽṹΪ

��������ͬ���칹�壬
�ʴ�Ϊ��3��


�����F�ķ���ʽC4H8O2��C��Dת��ΪF�ķ�Ӧ����������֪CΪ���ᣬDΪ�Ҵ���GΪ��ȩ��FΪ������������BCD�Ľṹ�����A�ķ���ʽ��Aת��ΪBCD�ķ�Ӧ���������Ƴ�AΪ

��
��1��BΪ

������ʽΪC9H10O3��A�Ľṹ��ʽ��

����Ӧ��Ϊ����ˮ�ⷴӦ��
�ʴ�Ϊ��C9H10O3��

��ˮ�⣻
��2����Ӧ��Ϊ������Ҵ���Ũ���������·���������Ӧ����Ӧ�ķ���ʽΪCH3COOH+C2H5OH
| Ũ���� |
| �� |
�ʴ�Ϊ��CH3COOH+C2H5OH
| Ũ���� |
| �� |
��3��GΪ��ȩ������-CHO������������Һ����������Ӧ����Ӧ�ķ���ʽΪCH3CHO+2Ag��NH3��2OH
| �� |
�ʴ�Ϊ��CH3CHO+2Ag��NH3��2OH
| �� |
��4��BΪ

����Ӧ��ͬ���칹���У��ٺ����ڶ�ȡ�������ṹ��
����B����ͬ�����ţ�˵��ҳ����-COOH��-OH��
�۲���FeCl3��Һ������ɫ��Ӧ��˵���������ǻ�������ܵĽṹΪ

��������ͬ���칹�壬
�ʴ�Ϊ��3��


��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ

 +2NaOH
+2NaOH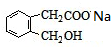 +CH3COONa+CH3CH2OH
+CH3COONa+CH3CH2OH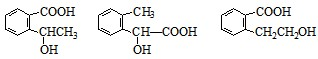 ������һ��
������һ�� ����CH2=CH2+H2O
����CH2=CH2+H2O ������ͼ�ش����⣺
������ͼ�ش����⣺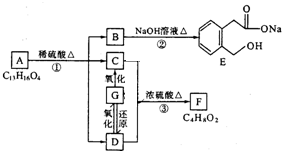 ͼ��A��B��C��D��E��F��G��Ϊ�л������B������ֻ��һ����״�ṹ������ͼʾ�ش����⣺
ͼ��A��B��C��D��E��F��G��Ϊ�л������B������ֻ��һ����״�ṹ������ͼʾ�ش����⣺