��Ŀ����
��֪A��B��C��D��EΪԭ������������������ֶ�����Ԫ�ء�����A��D��B��Eͬ���壬CԪ�ص��������������ڲ��������3����DԪ���ڵؿ��еĺ�������������D�ĵ��������õİ뵼����ϣ�EԪ�ص��������������۵Ĵ�����Ϊ2����ش�
��1��EԪ�ص��⻯���� ���ӣ�����ԡ��Ǽ��ԡ���������ʽ�� ��
��2��A��B��C����Ԫ���γɵ��⻯����۵��ɸߵ��͵�˳���� �����û�ѧʽ��ʾ��
��3��B��D��E����Ԫ�ص�����������ˮ�����������ǿ������˳���� �����û�ѧʽ��ʾ��
��4����ҵ����A�����Ʊ�D���ʵĻ�ѧ����ʽ�� ��
D��C�γɵĹ����������� ���塣
��1�����ԣ� ����2�֣���4�֣�
��2��H2O��NH3��CH4 ��2�֣�
��3��HNO3��H3PO4��H2SiO3 ��3�֣�
��4��SiO2+ 2C![]() Si+ 2CO����3�֣� ԭ�ӣ�3�֣�
Si+ 2CO����3�֣� ԭ�ӣ�3�֣�
����:
��

��ϰ��ϵ�д�
 �ʰ�Ӣ��ͬ����ϰ��ϵ�д�
�ʰ�Ӣ��ͬ����ϰ��ϵ�д� ѧϰʵ����ϵ�д�
ѧϰʵ����ϵ�д�
�����Ŀ
��֪A��B��C��D�ֱ���Cu��Ag��Fe��Al���ֽ����е�һ�֣���֪��A��C������ϡ���ᷴӦ�ų����壻��B��D�������η�Ӧ���û�������D����C��ǿ�Ӧ�ų����壬�ɴ˿����ƶ�A��B��C��D�����ǣ�������
| A��Fe��Cu��Al��Ag | B��Al��Cu��Fe��Ag | C��Cu��Ag��Al��Fe | D��Ag��Al��Cu��Fe |
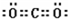
 ��֪A��B��C��D��E������������ͼ��ʾ��ת����ϵ�����ַ�Ӧ�P��Ӧ����δ�г���������ʱ��Ҫ�������������裩�������������о�����AԪ�أ�
��֪A��B��C��D��E������������ͼ��ʾ��ת����ϵ�����ַ�Ӧ�P��Ӧ����δ�г���������ʱ��Ҫ�������������裩�������������о�����AԪ�أ� ����ͨ������£���A��BΪ���ӣ�C��EΪ�����ӣ�DΪ�����ӣ����Ƕ�����10�����ӣ�B����A�����õ����ʿɵ����C��D��A��B��E��������Ӧ��ɵ�C��һ�ְ�ɫ��������ش�
����ͨ������£���A��BΪ���ӣ�C��EΪ�����ӣ�DΪ�����ӣ����Ƕ�����10�����ӣ�B����A�����õ����ʿɵ����C��D��A��B��E��������Ӧ��ɵ�C��һ�ְ�ɫ��������ش�