��Ŀ����
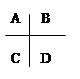
A��B��C��D��E��F�������ʵ�ת����ϵ��ͼ��ʾ(��Ӧ�����Ͳ��ֲ���δ���)��

(1)��AΪ�����ڽ������ʣ�DΪ�����ڷǽ������ʣ�������Ԫ�ص�ԭ������A��D��2��������Ԫ�ص�ԭ������������D��A��2����F��Ũ��Һ��A��D��Ӧ���к���ɫ�������ɣ���A��ԭ�ӽṹʾ��ͼΪ________����Ӧ�ܵĻ�ѧ����ʽΪ___________________________��
(2)��A�dz����ı�۽����ĵ��ʣ�D��F����̬���ʣ��ҷ�Ӧ����ˮ��Һ�н��С���Ӧ��Ҳ��ˮ��Һ�н��У������ӷ���ʽ��__________________________��
��֪����������D��F��Ӧ����B��д���÷�Ӧ�Ļ�ѧ����ʽ��___________________��
(3)��A��D��F���Ƕ����ڷǽ���Ԫ�ص��ʣ���A��D����Ԫ��ͬ���壬A��F����Ԫ��ͬ���ڣ���Ӧ�ٵĻ�ѧ����ʽΪ_____________________________________��

(1)��AΪ�����ڽ������ʣ�DΪ�����ڷǽ������ʣ�������Ԫ�ص�ԭ������A��D��2��������Ԫ�ص�ԭ������������D��A��2����F��Ũ��Һ��A��D��Ӧ���к���ɫ�������ɣ���A��ԭ�ӽṹʾ��ͼΪ________����Ӧ�ܵĻ�ѧ����ʽΪ___________________________��
(2)��A�dz����ı�۽����ĵ��ʣ�D��F����̬���ʣ��ҷ�Ӧ����ˮ��Һ�н��С���Ӧ��Ҳ��ˮ��Һ�н��У������ӷ���ʽ��__________________________��
��֪����������D��F��Ӧ����B��д���÷�Ӧ�Ļ�ѧ����ʽ��___________________��
(3)��A��D��F���Ƕ����ڷǽ���Ԫ�ص��ʣ���A��D����Ԫ��ͬ���壬A��F����Ԫ��ͬ���ڣ���Ӧ�ٵĻ�ѧ����ʽΪ_____________________________________��
(1) C��4HNO3(Ũ)==CO2����4NO2����2H2O
C��4HNO3(Ũ)==CO2����4NO2����2H2O
(2)2Fe2����Cl2== 2Fe3����2Cl�� H2��Cl2 ="=" 2HCl (3)2C��SiO2==Si��2CO��
 C��4HNO3(Ũ)==CO2����4NO2����2H2O
C��4HNO3(Ũ)==CO2����4NO2����2H2O (2)2Fe2����Cl2== 2Fe3����2Cl�� H2��Cl2 ="=" 2HCl (3)2C��SiO2==Si��2CO��
���������(1)��AΪ�����ڽ������ʣ�DΪ�����ڷǽ������ʣ���AΪMg��DΪC��FΪ���ᣬ����A��ԭ�ӽṹʾ��ͼΪ
 ����Ӧ�ܵĻ�ѧ����ʽΪC��4HNO3(Ũ)==CO2����4NO2����2H2O ��(2)��A�dz����ı�۽����ĵ��ʣ�D��F����̬���ʣ���AΪFe��DF�ֱ�Ϊ��������������Ӧ�ڵ����ӷ���ʽ��2Fe2����Cl2== 2Fe3����2Cl����D��F��Ӧ����B�Ļ�ѧ����ʽΪH2��Cl2 ="=" 2HCl ��(3)��A��D��F���Ƕ����ڷǽ���Ԫ�ص��ʣ���A��D��F�ֱ�ΪC��Si��O����Ӧ�ٵĻ�ѧ����ʽΪ2C��SiO2==Si��2CO����
����Ӧ�ܵĻ�ѧ����ʽΪC��4HNO3(Ũ)==CO2����4NO2����2H2O ��(2)��A�dz����ı�۽����ĵ��ʣ�D��F����̬���ʣ���AΪFe��DF�ֱ�Ϊ��������������Ӧ�ڵ����ӷ���ʽ��2Fe2����Cl2== 2Fe3����2Cl����D��F��Ӧ����B�Ļ�ѧ����ʽΪH2��Cl2 ="=" 2HCl ��(3)��A��D��F���Ƕ����ڷǽ���Ԫ�ص��ʣ���A��D��F�ֱ�ΪC��Si��O����Ӧ�ٵĻ�ѧ����ʽΪ2C��SiO2==Si��2CO����������������һ��ͼ�����費ͬ���������ó���ͬ�Ľ����������Ϣ���ܴ��õ���ȴ���࣬����ȽϷ�ʱ���ѶȽϴ�

��ϰ��ϵ�д�
 С��ʿ��ĩ����100��ϵ�д�
С��ʿ��ĩ����100��ϵ�д� ��У��ʦ������ҵ���Ӻ����Ծ�ϵ�д�
��У��ʦ������ҵ���Ӻ����Ծ�ϵ�д�
�����Ŀ
 ��Υ���� ԭ����
��Υ���� ԭ����
 H��
H�� H��
H�� H��
H�� Mg��
Mg�� Mg��
Mg�� Cu�й���______��Ԫ��_______��ԭ�ӣ�������������________����2311Na+ �е���������__ _����������_____�������������__ __��
Cu�й���______��Ԫ��_______��ԭ�ӣ�������������________����2311Na+ �е���������__ _����������_____�������������__ __��