��Ŀ����
��11�֣�25��ʱ������ƽ�ⳣ����
��ѧʽ CH3COOH H2CO3 HClO
����ƽ�ⳣ�� 1.8��10-5 K1 4.3��10-7 3.0��10-8
K25.6��10-11
�ش��������⣺
��1�����ʵ���Ũ��Ϊ0.1mol/L�������������ʣ�a.Na2CO3��bNaClO��c.CH3COONa
d.NaHCO3��PH�ɴ�С��˳���ǣ�_______________________�������ţ�
��2��������0.1mol/L��CH3COOH��Һ��ˮϡ���̣����б���ʽ������һ����С����:____________________��
A.c��H+�� B.c��H+��/c��CH3COOH�� C.c��H+����c��OH-�� D. c��OH-��/��H+��
��3�����Ϊ10mLPH=2�Ĵ�����Һ��һԪ��HX�ֱ��ˮϡ����1000mL��ϡ����PH�仯��ͼ����HX�ĵ���ƽ�ⳣ��_____������ڡ��������ڡ���С�ڡ��������ƽ�ⳣ����������_____________________________________________________________________��ϡ�ͺ�HX��Һ��ˮ���������c��H+��______������Һˮ�������c��H+��������ڡ��������ڡ���С�ڡ��������ǣ�_______________________________________________________��
��4��25��ʱ��CH3COOH��CH3COONa�Ļ����Һ������û��ҺPH=6������Һ��
c��CH3COO-��-c��Na+��=_______________________������ȷ��ֵ��
��1��a��b��d��c ��2��A
��3������ ϡ����ͬ������HX��PH�仯��CH3COOH������ǿ������ƽ�ⳣ���� ���� HX����ǿ��CH3COOH��ϡ�ͺ�c��H+��С��CH3COOH��Һ�е�c��H+�������Զ�ˮ����������������
��4��9.9��10-7mol/L��
����:

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д� ��2012?��ׯ��ģ��������ˮ��Һ�е���Ϊ����ѧ��ѧ����Ҫ���ݣ���֪�������ʵĵ��볣��ֵ��25�棩��
��2012?��ׯ��ģ��������ˮ��Һ�е���Ϊ����ѧ��ѧ����Ҫ���ݣ���֪�������ʵĵ��볣��ֵ��25�棩��
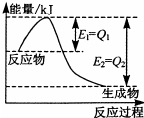 ���û�ѧ��Ӧԭ���о��������ȵȵ��ʼ��仯����ķ�Ӧ����Ҫ�����壮
���û�ѧ��Ӧԭ���о��������ȵȵ��ʼ��仯����ķ�Ӧ����Ҫ�����壮


