��Ŀ����
ij�о���ѧϰС����CO��ԭFe2O3��ʵ���У��ô����������ɵĺ�ɫ��ĩX��������ΪX��һ����Fe�����¶Ȳ���ʱ������Fe3O4��Ҳ�ܱ�����������Ϊ��̽��X����ɣ����ǽ���������ʵ�顣
I�����Լ���
| ʵ���� | ʵ����� | ʵ������ |
| �� | ȡ������ɫ��ĩX�����Թ�1�У�ע��Ũ���ᣬ�� | ��ɫ��ĩ���ܽ⣬��Һ�ʻ���ɫ�����������ݲ��� |
| �� | ��ȡ������ɫ��ĩX�����Թ�2�У�ע����������ͭ��Һ�������� | �м�������ɫ�������������н϶��ɫ����δ�ܽ� |
������ʵ�������ƶϣ���ɫ��ĩX�ijɷ��� ��
II�������ⶨ

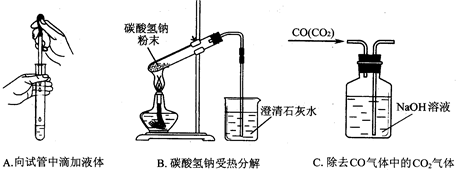
�� ����Z�������� ����ͼ19�������У������ճ���ʱ�����õ����� ������ĸ����

�� д����ҺY�еμ�H2O2��Һʱ������Ӧ�����ӷ���ʽ��
��
�� ����������ȣ������ڸ���������ȴ�����£���������ƽ����������Ϊb1 g���ٴμ��Ȳ���ȴ�����³���������Ϊb2 g����b1 �� b2 �� 0.3 g�����������Ӧ���еIJ�����
��
�� ��ͬѧ��Ϊ������������������H2O2���������費�䣬ֻҪ�ڿ����г�ַ����ԿɴﵽĿ�ġ����������ǣ��û�ѧ����ʽ��ʾ����
��
�� ͨ���������ݣ��ó�2.376 g��ɫ��ĩ�и��ɷֵ����ʵ���Ϊ ��
�� Fe �� Fe3O4
�� �� ���ˡ�ϴ�� (ֻѡ���˵�1��) A C E F
�� 2Fe2�� �� H2O2 �� 2H�� === 2Fe3�� 2H2O
�� �ٴμ��Ȳ����ڸ���������ȴ��������ֱ�����ε����������0.1gΪֹ��
�� 4Fe(OH)2 �� O2 �� 2H2O === 4Fe(OH)3
�� n(Fe) �� 0.001 mol n(Fe3O4) �� 0.01 mol
����:
��

| A��ʵ�������õ��ĵζ��ܡ�����ƿ����ʹ��ǰ����Ҫ��© | B�����ʵ��������60mL ��ϡ�������Һ������ʱӦѡ��100mL����ƿ | C������ƿ�к�����������ˮ���ᵼ���������Һ��Ũ��ƫС | D����ʽ�ζ���������ˮϴ�Ӻ�װ���Ũ�ȵ�ϡ���ᣬ���õ�NaOH��Һ��Ũ�Ƚ�ƫ�� | E��������Һʱ���������һ�ζ���ʱ���Ӷ������������ʵ����ƫ�� | F���к͵ζ�ʱ���������һ�ζ���ʱ���Ӷ������������ʵ����ƫ�� |