��Ŀ����
���и������ӣ�����ָ����Һ�д����������
[ ]
����ɫ��Һ�У�K+��Cl-��Na+��HCO3-��OH-
��ʹpH��ֽ������ɫ����Һ�У�K+��CO32-��Na+��AlO2-
��ˮ���������c(H+)=10-12 mol/L����Һ�У�ClO-��CO32-��NO3-��NH4+��SO32-
�ܼ���Mg�ܷų�H2����Һ�У�Mg2+��NH4+��Cl-��Na+��SO42-
����ʹ���ȱ�����Һ�У�MnO4-��NO3-��Na+��SO42-��Fe3+
��pH=0��Һ�У�Fe2+��Al3+��NO3-��Cl-
A���٢�
B���ڢܢ�
C���٢ڢ�
D���ۢ�
��ʹpH��ֽ������ɫ����Һ�У�K+��CO32-��Na+��AlO2-
��ˮ���������c(H+)=10-12 mol/L����Һ�У�ClO-��CO32-��NO3-��NH4+��SO32-
�ܼ���Mg�ܷų�H2����Һ�У�Mg2+��NH4+��Cl-��Na+��SO42-
����ʹ���ȱ�����Һ�У�MnO4-��NO3-��Na+��SO42-��Fe3+
��pH=0��Һ�У�Fe2+��Al3+��NO3-��Cl-
A���٢�
B���ڢܢ�
C���٢ڢ�
D���ۢ�
B

��ϰ��ϵ�д�
�����Ŀ
 ��Na+��HSO
��Na+��HSO ��PO
��PO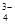 ��MnO
��MnO
 ��NO
��NO ��SO
��SO
 ��CO
��CO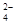 ��Cl
��Cl =0.1 mol/L����Һ�У�Na+��K+��SO
=0.1 mol/L����Һ�У�Na+��K+��SO