��Ŀ����
���и���������ָ����Һ�У����ܴ���������ǣ� ��
������������������ɫ��Һ�У�K+��NH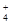 ��Na+��HSO
��Na+��HSO ��PO
��PO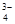 ��MnO
��MnO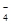
����ʹpH��ֽ������ɫ����Һ�У�K+��Na+��AlO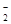 ��NO
��NO ��S
��S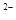 ��SO
��SO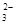
��ˮ�����H+Ũ��c(H+)=10-13mol��L-1����Һ�У�Cl ��CO
��CO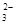 ��S2O
��S2O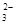 ��Fe3+��K+��Na+
��Fe3+��K+��Na+
�ܼ���Al�ܷų�H2����Һ�У�Na+��NH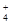 ��K+��SO
��K+��SO ��Cl
��Cl ��Br
��Br
��ʹʯ�������Һ�У�Fe3+��Na+��K+��NO ��SO
��SO ��Cl
��Cl
����������pH=1����Һ�У�Fe2+��Al3+��K+��Na+��NO ��I
��I ��Cl
��Cl ��S
��S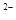
�ߺ��д���Fe3+����Һ��K+��NH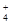 ��Na+��SCN
��Na+��SCN ��I
��I ��Br
��Br
�������£� =0.1 mol/L����Һ�У�Na+��K+��SO
=0.1 mol/L����Һ�У�Na+��K+��SO ��SiO
��SiO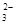 ��NO
��NO
A���٢ۢܢ� B���٢ۢޢ� C���ڢܢݢ� D���ڢݢߢ�
C
��������
�����������MnO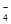 ����ɫ�ģ�����ʹpH��ֽ������ɫ����Һ�Ǽ�����Һ����Щ�����ڼ��������¿��Թ��棻��ˮ�����H+Ũ��c(H+)=10-13mol��L-1����Һ����ˮ�ĵ����ܵ������ƣ�˵����Һ���������ԣ�Ҳ�����Ǽ��Ե���Һ������������CO
����ɫ�ģ�����ʹpH��ֽ������ɫ����Һ�Ǽ�����Һ����Щ�����ڼ��������¿��Թ��棻��ˮ�����H+Ũ��c(H+)=10-13mol��L-1����Һ����ˮ�ĵ����ܵ������ƣ�˵����Һ���������ԣ�Ҳ�����Ǽ��Ե���Һ������������CO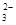 ��S2O
��S2O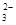 �����ܹ��棬����������Fe3+���ܹ��棻�ܼ���Al�ܷų�H2����Һ������������Һ��Ҳ�����Ǽ�����Һ������������NH
�����ܹ��棬����������Fe3+���ܹ��棻�ܼ���Al�ܷų�H2����Һ������������Һ��Ҳ�����Ǽ�����Һ������������NH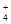 �����ܹ��棻��ʹʯ�������Һ��������Һ�����Թ��棻��NO
�����ܹ��棻��ʹʯ�������Һ��������Һ�����Թ��棻��NO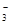 ����������������ǿ�������ԣ���������Fe2+��I
����������������ǿ�������ԣ���������Fe2+��I ������˲��ܹ��棻�ߺ��д���Fe3+����Һ��������SCN
������˲��ܹ��棻�ߺ��д���Fe3+����Һ��������SCN ��I
��I ���棻�������£�
���棻�������£� 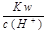 =0.1 mol/L����Һ�У����������ӵ�Ũ��Ϊ0.1 mol/L��Ҳ����һ��������Һ�����Թ��档����������ѡC��
=0.1 mol/L����Һ�У����������ӵ�Ũ��Ϊ0.1 mol/L��Ҳ����һ��������Һ�����Թ��档����������ѡC��
���㣺���ӹ�����й�֪ʶ��

 ���ɶ���ܲ��¿�ֱͨ�п�ϵ�д�
���ɶ���ܲ��¿�ֱͨ�п�ϵ�д�