��Ŀ����
��������ʱ������(N2H4)Ϊȼ�ϣ��ö�������Ϊ�����������������ʷ�Ӧ���ɵ�����ˮ��������֪��N2(g)+2O2(g)====2NO2(g)����H1=+67.7 kJ��mol-1
N2H4(g)+O2(g)====N2(g)+2H2O(g)����H2=-534 kJ��mol-1
�Լ���1 mol�ºͶ���������ȫ��Ӧʱ�ų�������Ϊ_________��д���������������Ӧ���Ȼ�ѧ����ʽ______________________________________��
�������������֪��
N2(g)+2O2(g)====2NO2(g)����H1=+67.7 kJ��mol-1 ��
N2H4(g)+O2(g)====N2(g)+2H2O(g)����H2=-534 kJ��mol-1 ��
���ݸ�˹���ɣ�Ϊ�ڷ�Ӧ������ȥO2���õ�N2H4��NO2�ķ�Ӧ����ڡ�2-�ٵã�
2N2H4(g)-N2(g)====2N2(g)+4H2O(g)-2NO2(g)����H=2��H2-��H1�������ã�
2N2H4(g)+2NO2(g)====3N2(g)+4H2O(g)����H
��H=2��H2-��H1=534 kJ��mol-1��2-67.7 kJ��mol-1=-1135.7 kJ��mol-1��
��1 mol N2H4��Ӧ�ų�������Ϊ567.85 kJ
�𰸣�567.85��
2N2H4(g)+2NO2(g)====3N2(g)+4H2O(g)����H=-1135.7 kJ��mol-1��

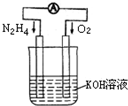 ���û�ѧ��Ӧԭ���о��������ȵ��ʼ��仯����ķ�Ӧ����Ҫ���壮
���û�ѧ��Ӧԭ���о��������ȵ��ʼ��仯����ķ�Ӧ����Ҫ���壮