��Ŀ����
3����5.6g Feȫ������200mLһ��Ũ�ȵ�������Һ�У��õ���״���µ�����2.24L���ֲ�÷�Ӧ����Һ��H+��Ũ��Ϊ0.2mol•L-1���跴Ӧǰ����Һ����ı仯���Բ��ƣ�������˵������ȷ���ǣ�������| A�� | 2.24 L������NO | |
| B�� | ��Ӧ��Feȫ��ת��ΪFe3+ | |
| C�� | ��Ӧ�����Һ��c��NO3-��=1.7 mol•L-1 | |
| D�� | ��Ӧ�����Һ�������ܽ�2.24 g Fe |
���� n��Fe��=$\frac{5.6g}{56g/mol}$=0.1mol��n�����壩=$\frac{2.24L}{22.4L/mol}$=0.1mol����÷�Ӧ����Һ��H+��Ũ��Ϊ0.2mol•L-1��������������ɵ����غ��֪��Feʧȥ0.3mol����ʱ��Nԭ�ӵõ�0.3mol���ӣ�����������ΪNO���Դ������
��� �⣺n��Fe��=$\frac{5.6g}{56g/mol}$=0.1mol��n�����壩=$\frac{2.24L}{22.4L/mol}$=0.1mol����÷�Ӧ����Һ��H+��Ũ��Ϊ0.2mol•L-1�������������
A���ɵ����غ��֪��0.1mol����3-0��=0.1mol����5-x�������x=2��������ΪNO����A��ȷ��
B�������������Ӧ����Һ�к���Fe3+����B��ȷ��
C����Nԭ���غ��֪��c��NO3-��=$\frac{0.1mol��3+0.2L��0.2mol/L}{0.2L}$=1.7mol/L����C��ȷ��
D��ԭ��������ʵ���Ϊ0.1mol��3+0.2L��0.2mol/L+0.1mol=0.44mol����3Fe+8HNO3=3Fe��NO3��2+2NO��+4H2O��֪���������FeΪ0.44mol��$\frac{3}{8}$��56g/mol=9.24g�����������ܽ�9.24-5.6=3.64g����D����
��ѡD��
���� ���⿼��������ԭ��Ӧ�ļ��㣬Ϊ��Ƶ���㣬������������������غ㡢ԭ���غ��Ӧ��Ϊ���Ĺؼ������ط�������������Ŀ��飬��Ŀ�Ѷ��еȣ�

 Ӧ�����������Ĵ���ѧ������ϵ�д�
Ӧ�����������Ĵ���ѧ������ϵ�д�| A�� | ���ع��ͻ��ռӹ�Ϊ������ͣ������Դ�������� | |
| B�� | ��˿����������ø��������ˮ�����ɵõ���-������ | |
| C�� | ������������õ�����������Ҷľ��ԭ�ϵ���Ҫ�ɷ�����ά�� | |
| D�� | �ߴ��ȵľ��������Ʊ�����ܵȰ뵼�塢����оƬ |
| A�� | �߲˺ʹ����и�����ά�أ���ά����������ø����������ˮ��������� | |
| B�� | ���������кͷ�����������������ԭ���ǹ�ҵ������ˮ���õķ��� | |
| C�� | ��ֻ�õ�����Һ����ʳ�����Ƿ�ӵ� | |
| D�� | ��������Һ�м��˱���Na2SO4��Һ���г�����������Ϊ�����ʱ��� |
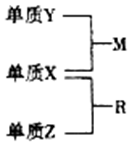 X��Y��Z�Ƕ�����Ԫ�أ�ԭ�Ӱ뾶�Ĵ�С��r��Y����r��X����r��Z��������Ԫ�ص�ԭ������֮��Ϊ15��X��Y��Z����Ԫ�صij����������ʵ������¿ɷ�����ͼ��ʾת����ϵ������RΪ10���ӷ��ӣ���һ�ֳ��������ܼ�������˵������ȷ���ǣ�������
X��Y��Z�Ƕ�����Ԫ�أ�ԭ�Ӱ뾶�Ĵ�С��r��Y����r��X����r��Z��������Ԫ�ص�ԭ������֮��Ϊ15��X��Y��Z����Ԫ�صij����������ʵ������¿ɷ�����ͼ��ʾת����ϵ������RΪ10���ӷ��ӣ���һ�ֳ��������ܼ�������˵������ȷ���ǣ�������| A�� | XԪ��λ�����ڱ��еĵ�16�� | |
| B�� | X���⻯��е�һ������Y���⻯��ķе� | |
| C�� | X��Y��ZԪ�ؿ������һ�ַ���ʽΪZ2Y2X4�Ļ������ˮ��Һ��ˮ�ĵ����ܵ������� | |
| D�� | X��Y��ZԪ������֮������γ�ԭ�Ӹ�����Ϊ1��1�Ļ����� |
| A�� | ���Ȼ�����Һ�м��������ˮ��Al3++3NH3•H2O�TAl��OH��3��+3NH4+ | |
| B�� | ��Ͷ��ˮ�У�Na+H2O�TNa++2OH-+H2�� | |
| C�� | �������������������Һ��Ӧ��2Cl2+2OH-�T3Cl-+ClO-+H2O | |
| D�� | ʯ��ʯ����������Һ�У�2H++CO32-�TCO2��+H2O |
| A�� | �����ƹ�ũ�����������μ������Լ���ũҩ��ʹ�� | |
| B�� | ʹ�ü�øϴ�·ۣ�ˮ��Խ�ߣ�ϴ��Ч������ | |
| C�� | ��CO2�ϳɾ�̼�����ɽ������ϣ�����ʵ�֡�̼����ѭ������ | |
| D�� | ����ԺҪ�������Ƹ�����ˮ�ࡢ��̿�ȸߺ�����ҵ��������������ʵ����̼���á��ľ���ٴ� |
 ����Ӧ����ȡ����Ӧ��
����Ӧ����ȡ����Ӧ��