��Ŀ����
�����ӵ�����ԼΪ6.02��1023mol-1������˵����һ����ȷ����
A��1 L 1 mol•L-1��HAc��Һ�к�����������ԼΪ6.02��1023
B��18 g D2O����ˮ����ȫ��⣬ת�Ƶĵ�����ԼΪ6.02��1023
C����̬�£�22.4L NO2���壬������ԭ����ԼΪ2��6.02��1023
D����̬�£�14g��ϩ�ͱ�ϩ�Ļ�����У����й��õ��Ӷ���ĿԼΪ3��6.02��1023
D
��������
���������A��������������ڵ���ƽ�⣬���1L 1mol•L-1��HAc��Һ�к�����������С��6.02��1023����A����B��18g D2O����ˮ�����ʵ�����18g��20g/mol��0.9mol����ˮ��ȫ��⣬����ݷ���ʽ2D2O��2D2+O2��֪��ת�Ƶĵ�����ԼΪ0.9��2��6.02��1023����B����C��������������������������ڻ�ѧƽ��2NO2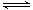 N2O4����̬�£�22.4L NO2�������ʵ���Ϊ1mol��������ԭ��������2��6.02��1023��С��4��6.02��1023����C����D����ϩ�ͱ�ϩ�����ʽ����CH2������14g��ϩ�ͱ�ϩ�Ļ�����У����й��õ��Ӷ���ĿԼΪ
N2O4����̬�£�22.4L NO2�������ʵ���Ϊ1mol��������ԭ��������2��6.02��1023��С��4��6.02��1023����C����D����ϩ�ͱ�ϩ�����ʽ����CH2������14g��ϩ�ͱ�ϩ�Ļ�����У����й��õ��Ӷ���ĿԼΪ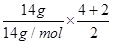 ��6.02��1023��3��6.02��1023����D��ȷ����ѡD��
��6.02��1023��3��6.02��1023����D��ȷ����ѡD��
���㣺���鰢���ӵ��������йؼ�����ж�

 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�| A��4 g��ˮ��D2O��������������Ϊ0.2��6.02��1023 | B��2.8 g��ϩ�ͱ�ϩ�Ļ������������̼ԭ����Ϊ0.2��6.02��1023 | C��4.48 L H2��O2�Ļ������������������Ϊ0.2��6.02��1023 | D��0.2 mol Cl2�ܽ��ڵ������ˮ�У�ת�Ƶ�����Ϊ0.2��6.02��1023 |
 �����ӵ�����ԼΪ6.02��1023mol-1������˵����һ����ȷ���ǣ�������
�����ӵ�����ԼΪ6.02��1023mol-1������˵����һ����ȷ���ǣ�������