��Ŀ����
��֪ij�¶�ʱCH3COOH�ĵ���ƽ�ⳣ��ΪK�����¶�����20 mL 0.1 mol��L��1 CH3COOH��Һ����μ���0.1 mol��L��1 NaOH��Һ����pH�仯������ͼ��ʾ(�����¶ȱ仯)������˵���в���ȷ���ǣ� ��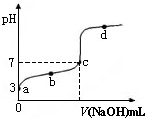
| A��a��ʱ��CH3COOH�ĵ������1% |
| B��b��ʱ����Һ��c(CH3COO��)��c(Na��) |
| C��c���ʾCH3COOH��NaOHǡ�÷�Ӧ��ȫ |
D��b��d���ʾ����Һ�� ������K ������K |
C
�������������A��a����c��H+����10��3mol/L�����ڴ���Ϊ���ᣬ��������ˮ�ĵ��룬����ĵ���ԶԶ����ˮ�ĵ��룬������Һ��������Ũ�Ƚ��Ƶ��ڴ��������Ũ�ȣ���CH3COOH�ĵ������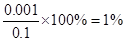 ����A��ȷ��
����A��ȷ��
B�����ݵ���غ��֪����Һ����c��Na+��+c��H+����c��OH-��+c��CH3COO-����b��ʱ��c��H+����c��OH����������c��CH3COO������c��Na+������B��ȷ��
C��������������Ʒ�Ӧ���ɴ����ƣ���������ǿ����������ˮ��Һ�ʼ��ԣ������ǡ�÷�Ӧʱ��ҺӦ�óʼ��ԣ���C����Һ�����ԣ�˵�����������C����
D��b��d������Һ���¶���ͬ������b��d���ʾ����Һ�� ������K����D��ȷ����ѡC��
������K����D��ȷ����ѡC��
���㣺��������Ϻ���Һʶ���Ե��ж�
�����������Ǹ߿��еij������ͣ������е��Ѷȵ����⡣���ض�ѧ������������������������ѧ������˼ά�����ͷ�ɢ˼ά����������ʱע��ƽ�ⳣ��Kֻ���¶��йأ��������κ��������ء�

 ��ҵ����ϵ�д�
��ҵ����ϵ�д� ͬ��ѧ��һ�ζ���ϵ�д�
ͬ��ѧ��һ�ζ���ϵ�д� ��֪ij�¶�ʱCH3COOH�ĵ���ƽ�ⳣ��ΪK�����¶�����20mL0.1mol/LCH3COOH��Һ����μ���0.1mol/L NaOH��Һ����pH�仯������ͼ��ʾ�������¶ȱ仯��������˵���в���ȷ���ǣ�������
��֪ij�¶�ʱCH3COOH�ĵ���ƽ�ⳣ��ΪK�����¶�����20mL0.1mol/LCH3COOH��Һ����μ���0.1mol/L NaOH��Һ����pH�仯������ͼ��ʾ�������¶ȱ仯��������˵���в���ȷ���ǣ������� ��2009?���ݶ�ģ����֪ij�¶�ʱCH3COOH�ĵ���ƽ�ⳣ��ΪK�����¶�����20mL 0.1mol?L-1 CH3COOH��Һ����μ���0.1mol?L-1 NaOH��Һ����pH�仯������ͼ��ʾ�������¶ȱ仯��������˵���в���ȷ���ǣ�������
��2009?���ݶ�ģ����֪ij�¶�ʱCH3COOH�ĵ���ƽ�ⳣ��ΪK�����¶�����20mL 0.1mol?L-1 CH3COOH��Һ����μ���0.1mol?L-1 NaOH��Һ����pH�仯������ͼ��ʾ�������¶ȱ仯��������˵���в���ȷ���ǣ������� ��2010?������ģ�⣩��֪ij�¶�ʱCH3COOH�ĵ���ƽ�ⳣ��ΪK�����¶��£���0.1000mol?L-1NaOH��Һ�ζ�20.00mL 0.1000mol?L-1CH3COOH��Һ���õζ�������ͼ�������¶ȱ仯��������˵���в���ȷ���ǣ�������
��2010?������ģ�⣩��֪ij�¶�ʱCH3COOH�ĵ���ƽ�ⳣ��ΪK�����¶��£���0.1000mol?L-1NaOH��Һ�ζ�20.00mL 0.1000mol?L-1CH3COOH��Һ���õζ�������ͼ�������¶ȱ仯��������˵���в���ȷ���ǣ�������
 ������K
������K