��Ŀ����
18��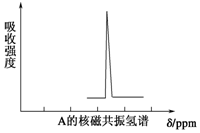 ��һ���ºϳɵĸ߶ȶԳƵ��л���A����֪10.4g A������O2�г��ȼ�գ������ɵĻ����������ͨ��������Ũ����ͼ�ʯ�ң��ֱ�����7.2g��35.2g��������ʣ������ΪO2��
��һ���ºϳɵĸ߶ȶԳƵ��л���A����֪10.4g A������O2�г��ȼ�գ������ɵĻ����������ͨ��������Ũ����ͼ�ʯ�ң��ֱ�����7.2g��35.2g��������ʣ������ΪO2����1��A��ʵ��ʽΪCH
��2����ȡ10.4g A������ʹ�������������ܶ�����ͬ������H2��52������A�ķ���ʽ��C8H8
��3��A���ӵĺ��������ʾ�����ͼ���A���ӵĺ˴Ź���������ͼ����A�ļ���ʽ��

��4��ͬʱ����������������A��ͬ���칹��ڷ����廯�����ֻ��һ���������л���Ľṹ��ʽ
 ����˴Ź���������5�����շ壮
����˴Ź���������5�����շ壮
���� ��1������Ũ��������7.2gΪˮ����������ʯ������35.2gΪ������̼�����������������غ��Ƿ�����Ԫ�أ����Ӷ���ȷ����ʵ��ʽ��
��2������ܶ�����Է������������ȣ��ݴ˿�ȷ��A����Է���������Ȼ������ʵ��ʽȷ�������ʽ��
��3��A���ӵĺ��������ʾ�����ͼ���˵��A����̼��������A���ӵĺ˴Ź�������֪A�������к���1��H���ݴ˿�ȷ����ṹ��
��4����A��ͬ���칹�壬��A������ͬ����ʽ���ڷ����廯��������к���1����������ֻ��һ���������ݴ˿�ȷ�������������л���Ľṹ��ʽ����ϸýṹ��ʽ�к��е�ЧH����Ŀ�ж���˴Ź����������շ壮
��� �⣺��1��7.2gˮ�����ʵ���Ϊ��$\frac{7.2g}{18g/mol}$=0.4mol��n��H��=0.8 mol��35.2g������̼�����ʵ���Ϊ��$\frac{35.2g}{44g/mol}$=0.8mol��n��C��=n��CO2��=0.8 mol�����л����к���C��H��������Ϊ��12g/mol��0.8mol+1g/mol��0.8mol=10.4g��˵�����л�������в�������Ԫ�أ��������C��Hԭ�ӵ����ʵ���֮��=0.8mol��0.8mol=1��1������ʵ��ʽΪCH��
�ʴ�Ϊ��CH��
��2��A����Է�������Ϊ��2��52=104���������ʽΪ��CH��n����13n=104��n=8����A�ķ���ʽΪC8H8��
�ʴ�Ϊ��C8H8��
��3��A���ӵĺ��������ʾ�����ͼ���˵��A����̼��������A�ĺ˴Ź�������ͼ��֪��A�������к���1��Hԭ�ӣ����A�Ǹ߶ȶԳƵ��л����֪��A�������飬�����ʽΪ�� ��
��
�ʴ�Ϊ�� ��
��
��4��ͬʱ����������������A��ͬ���칹�壬����ʽΪC8H8���ڷ����廯��������к��б�������ֻ��һ��������ʣ���2��C���1�����ţ��������������л���Ľṹ��ʽΪ�� ��������к���5�ֵ�ЧHԭ�ӣ�����˴Ź��������к���5�����շ壬
��������к���5�ֵ�ЧHԭ�ӣ�����˴Ź��������к���5�����շ壬
�ʴ�Ϊ�� ��5��
��5��
���� ���⿼���л���ṹ�����ʣ���Ŀ�Ѷ��еȣ��ƶ�A�ķ���ʽΪ���ؼ���ע���������ճ����л�����ɡ��ṹ�����ʣ���4��Ϊ�״��㣬����������ѧ�����ۺ�Ӧ��������

| A�� | ԭ�Ӱ뾶��d��c��b��a | |
| B�� | d���������ˮ������ǿ�� | |
| C�� | c��������������ᷴӦ������Ӧ | |
| D�� | d���ʵ������Ա�a���ʵ�������ǿ |
| A�� | ʹ���������ܼӴ�Ӧ���� | |
| B�� | �ı䷴Ӧ���ѹǿ�Է�Ӧ���ʲ���Ӱ�� | |
| C�� | �������µͣ���Ӧ���ʽ��ͣ�������Σ������ | |
| D�� | ����������������ı䣬���Դ˻�ѧ��Ӧ��������Ӱ�� |
 =CH-C��C-CF3�д���ͬһƽ���ڵ�ԭ���������ǣ�������
=CH-C��C-CF3�д���ͬһƽ���ڵ�ԭ���������ǣ�������| A�� | 12�� | B�� | 14�� | C�� | 18�� | D�� | 20�� |
| A�� | ��16 g��ԭ�ӵĶ�����̼�����к��еĦļ���ĿΪNA | |
| B�� | 3.9 g Na2O2�������0.15NA������ | |
| C�� | ���³�ѹ�£�5 g D2O���е�������������������������Ϊ2.5NA | |
| D�� | 2 mol SO2��1 mol O2��һ�������·�Ӧ���û�����������С��2NA |
| A�� | Na�ĺ����������Ǽ��������� | |
| B�� | �Ӻ�ˮ����ȡ������Ԫ�صĵ��ʵĹ��̶��漰������ԭ��Ӧ | |
| C�� | ������Ԫ���Ի���̬����ʱ�γɵĻ�ѧ��ֻ�����Ӽ� | |
| D�� | ��������NaCl��MgCl2��Һʱ���Եõ�����NaCl��MgCl2 |
| A�� | ����ˮ������������������ڽ��� | |
| B�� | ����������������ɢϵ�ı��������Ƿ�ɢ������ֱ����С��1-100nm֮�� | |
| C�� | ��ƽ�й�����NaCl��Һ��Fe��OH��3����ʱ������������ͬ | |
| D�� | Fe��OH��3�����ܹ�ʹˮ��������Ĺ�������������ﵽ��ˮ��Ŀ�� |

| A�� | ��ͼװ���ñ�Һ�ζ�����Һ | B�� | ����ͼװ���Ʊ�Fe��OH��2 | ||
| C�� | ��ͼװ�ÿ��Ƶý����� | D�� | ��ͼ��֤NaHCO3��Na2CO3���ȶ��� |