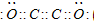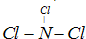��Ŀ����
��15�֣�����A��B��C��D��E����Ԫ��ԭ��������������,ֻ��E���Ƕ�����Ԫ��;����ֱ��������±���
Ԫ�ر�� | Ԫ������ |
A | ���ڱ���ԭ�Ӱ뾶��С |
B | ���⻯���������� |
C | ������������������������Ӳ������ |
D | ����ϼ�Ϊ+7 |
E | �������Ӧ����㷺�Ľ��� |
��1��Ԫ��D�����������Ϊ��ɫҺ�壬��������һ����ˮ��ϵõ�һ��ϡ��Һ��д���÷�Ӧ�Ļ�ѧ����ʽ_______________________ ��
��2����һ����ѧ����ʽ��ʾC��E�Ľ�����ǿ��_______��
��3��A��B����Ԫ�����һ�������ӣ�����ij��Һ�к��и����ӵķ����ǣ�_________________��
��4����C��ij�������������ˮ���������ʵ�ˮ��Һ�����ԣ���ԭ����_________________���������ӷ���ʽ��ʾ��
��5��E�����ں�ˮ���ױ���ʴ��д��������Ӧʽ��_________________
��1��Cl2O7+H2O=2HClO4
��2��2Al+Fe2O3=Al2O3+2Fe
��3��ȡ��������Һ���Թ��У��μ�Ũ����������Һ���ȣ���ʪ��ĺ�ɫʯ����ֽ�����Թܿڣ���ֽ��졣
��4��Al3++3H2O=Al(OH)3+3H+
��5��O2+2H2O+4e-=4OH-

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
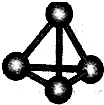 ����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��
����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��