��Ŀ����
ʵ��������Ҫ22.4l����״����SO2���壮��ѧС��ͬѧ���ݻ�ѧ����ʽZn+2H2SO4��Ũ��  ZnSO4+SO2��+2H2O�����ȡ65.0gп����98%��ŨH2SO4����=1.84g?cm-3��110mL��ַ�Ӧпȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ������ʣ�
ZnSO4+SO2��+2H2O�����ȡ65.0gп����98%��ŨH2SO4����=1.84g?cm-3��110mL��ַ�Ӧпȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ������ʣ�
��1����ѧС�����Ƶõ������л��е���Ҫ�������������______�������ʽ�����������ֽ������Ҫԭ����______
���û�ѧ����ʽ�ͱ�Ҫ�����ּ���˵����
��2��Ϊ֤ʵ��ط�������ѧС���ͬѧ�����ʵ�飬��װ������װ�ã�������ȡ���������̽����
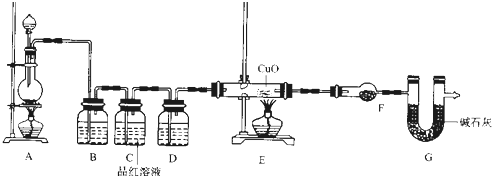
��װ��B�м�����Լ�______��������______��
��װ��D������Լ�______��װ��F������Լ�______��
�ۿ�֤ʵһ������п����һ������Ũ���ᷴӦ�����ɵ������л���ij���������ʵ��������______��
��U��G������Ϊ______��
�⣺��1����������Ũ�Ƚ��ͣ�п��ϡ���ᷴӦ�������������Ի�ѧС�����Ƶõ������л��е���Ҫ��������������������ʴ�Ϊ��H2�����ŷ�Ӧ�Ľ��У�����Ũ�Ƚ��ͣ���ʹп��ϡ���ᷴӦ����H2��Zn+H2SO4�TZnSO4+H2����
��2����Bװ���dz�ȥ�������������������Ƶȼ�Һ���գ��ʴ�Ϊ��NaOH��Һ����ȥ��������е�SO2��
��Dװ����Ũ���ᣬ��ˮ�������ã�Fװ����ˮ����ͭ�������Ƿ���ˮ���ɣ��ʴ�Ϊ��Ũ�����ˮ����ͭ��
��Eװ�����û�ԭ�����廹ԭ����ͭ��Fװ����ˮ����ͭ�������Ƿ���ˮ���ɣ�ֻҪ��ɫ���ɫ����ˮ����ͭ����ɫ����֤�����������ʴ�Ϊ��װ��E�в������к�ɫCuO��ĩ���ɫ�������F����ˮ����ͭ����ɫ��
��G�Ƿ�ֹ������H2O�������ܶ�Ӱ����������ļ��飬�ʴ�Ϊ����ֹ������H2O�������ܶ�Ӱ����������ļ��飮
��������1����������Ũ�Ƚ��ͣ�п��ϡ���ᷴӦ����������
��2��A����ȡ�����װ�ã��Ʊ����������ж�������������ˮ������Bװ���dz�ȥ�������������������Ƶȼ�Һ���գ�Cװ���Ǽ�����������Ƿ������Dװ����Ũ���ᣬ��ˮ�������ã�Eװ�����û�ԭ�����廹ԭ����ͭ��Fװ����ˮ����ͭ�������Ƿ���ˮ���ɣ�G�Ƿ�ֹ������H2O�������ܶ�Ӱ����������ļ��飮
������������ʵ��̽���⣬�����������ȡ��������ij��Ӻ���������գ���������ļ��飬�ۺ��ԱȽ�ǿ���Ѷ��еȣ�
��2����Bװ���dz�ȥ�������������������Ƶȼ�Һ���գ��ʴ�Ϊ��NaOH��Һ����ȥ��������е�SO2��
��Dװ����Ũ���ᣬ��ˮ�������ã�Fװ����ˮ����ͭ�������Ƿ���ˮ���ɣ��ʴ�Ϊ��Ũ�����ˮ����ͭ��
��Eװ�����û�ԭ�����廹ԭ����ͭ��Fװ����ˮ����ͭ�������Ƿ���ˮ���ɣ�ֻҪ��ɫ���ɫ����ˮ����ͭ����ɫ����֤�����������ʴ�Ϊ��װ��E�в������к�ɫCuO��ĩ���ɫ�������F����ˮ����ͭ����ɫ��
��G�Ƿ�ֹ������H2O�������ܶ�Ӱ����������ļ��飬�ʴ�Ϊ����ֹ������H2O�������ܶ�Ӱ����������ļ��飮
��������1����������Ũ�Ƚ��ͣ�п��ϡ���ᷴӦ����������
��2��A����ȡ�����װ�ã��Ʊ����������ж�������������ˮ������Bװ���dz�ȥ�������������������Ƶȼ�Һ���գ�Cװ���Ǽ�����������Ƿ������Dװ����Ũ���ᣬ��ˮ�������ã�Eװ�����û�ԭ�����廹ԭ����ͭ��Fװ����ˮ����ͭ�������Ƿ���ˮ���ɣ�G�Ƿ�ֹ������H2O�������ܶ�Ӱ����������ļ��飮
������������ʵ��̽���⣬�����������ȡ��������ij��Ӻ���������գ���������ļ��飬�ۺ��ԱȽ�ǿ���Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
ʵ��������Ҫ22��4 l����״����SO2���塣��ѧС��ͬѧ���ݻ�ѧ����ʽZn+
�� 2H2SO4��Ũ����![]() ���� ZnSO4+SO2��+2H2O�����ȡ65.0gп����98%��ŨH2SO4��
���� ZnSO4+SO2��+2H2O�����ȡ65.0gп����98%��ŨH2SO4��![]() ��110mL��ַ�Ӧпȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ������ʡ�
��110mL��ַ�Ӧпȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ������ʡ�
�� ��1����ѧС�����Ƶõ������л��е���Ҫ������������������������� �������ʽ�����������ֽ������Ҫԭ������������������������������������������������
�� ���û�ѧ����ʽ�ͱ�Ҫ�����ּ���˵����
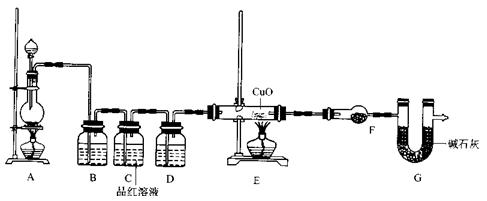
�� ��2��Ϊ֤ʵ��ط�������ѧС���ͬѧ�����ʵ�飬��װ������װ�ã�������ȡ���������̽����
|
��װ��B�м�����Լ��� ������ ���������������������������� ��
��װ��D������Լ��������� ��װ��F������Լ��������������������� ��
�ۿ�֤ʵһ������п����һ������Ũ���ᷴӦ�����ɵ������л���ij���������ʵ���������������������������������������������� ��
��U��G�������������������������������������������� .


 ZnSO4+SO2��+2H2O�����ȡ65.0gп����98%��ŨH2SO4��
ZnSO4+SO2��+2H2O�����ȡ65.0gп����98%��ŨH2SO4�� ��110mL��ַ�Ӧпȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ������ʡ�
��110mL��ַ�Ӧпȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ������ʡ�
