��Ŀ����
ʵ��������Ҫ22.4 L(��״��)SO2���壮��ѧС��ͬѧ���ݻ�ѧ����ʽZn��2H2SO4(Ũ)![]() ZnSO4��SO2����2H2O�����ȡ65.0 gп����98����ŨH2SO4(�ѣ�1.84 g��cm��3)110 mL��ַ�Ӧ��пȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ�������
ZnSO4��SO2����2H2O�����ȡ65.0 gп����98����ŨH2SO4(�ѣ�1.84 g��cm��3)110 mL��ַ�Ӧ��пȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ�������
(1)��ѧС��������Ƶõ������л��е���Ҫ�������������________(�����ʽ)��
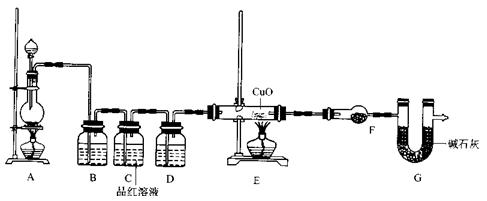
(2)Ϊ֤ʵ��ط�������ѧС���ͬѧ�����ʵ�飬��װ����װ�ã�������ȡ���������̽����

��װ��B�м�����Լ�________��������________��
��װ��D�м�����Լ�________��װ��F�м�����Լ�________��
�ۿ�֤ʵһ������п����һ������Ũ���ᷴӦ�����ɵ������л���ij���������ʵ��������________��
��U��G��������________��
������
����(2)��NaOH��Һ(��KMnO4������������Ҳ����)(1��)����ȥ��������е�SO2(1��)
������Ũ����(1��)����ˮ����ͭ(1��)
������װ��E�в������к�ɫCuO��ĩ���ɫ�������F����ˮ����ͭ����ɫ(1��)
�����ܷ�ֹ������H2O�������ܶ�Ӱ����������ļ���(1��)

 ��У����ϵ�д�
��У����ϵ�д�ʵ��������Ҫ22��4 l����״����SO2���塣��ѧС��ͬѧ���ݻ�ѧ����ʽZn+
�� 2H2SO4��Ũ����![]() ���� ZnSO4+SO2��+2H2O�����ȡ65.0gп����98%��ŨH2SO4��
���� ZnSO4+SO2��+2H2O�����ȡ65.0gп����98%��ŨH2SO4��![]() ��110mL��ַ�Ӧпȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ������ʡ�
��110mL��ַ�Ӧпȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ������ʡ�
�� ��1����ѧС�����Ƶõ������л��е���Ҫ������������������������� �������ʽ�����������ֽ������Ҫԭ������������������������������������������������
�� ���û�ѧ����ʽ�ͱ�Ҫ�����ּ���˵����
�� ��2��Ϊ֤ʵ��ط�������ѧС���ͬѧ�����ʵ�飬��װ������װ�ã�������ȡ���������̽����
|
��װ��B�м�����Լ��� ������ ���������������������������� ��
��װ��D������Լ��������� ��װ��F������Լ��������������������� ��
�ۿ�֤ʵһ������п����һ������Ũ���ᷴӦ�����ɵ������л���ij���������ʵ���������������������������������������������� ��
��U��G�������������������������������������������� .

 ZnSO4+SO2��+2H2O�����ȡ65.0gп����98%��ŨH2SO4��
ZnSO4+SO2��+2H2O�����ȡ65.0gп����98%��ŨH2SO4�� ��110mL��ַ�Ӧпȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ������ʡ�
��110mL��ַ�Ӧпȫ���ܽ⣬�����Ƶõ����壬��ͬѧ��Ϊ���ܻ������ʡ�
