��Ŀ����
��֪�����£�Ksp[Mg(OH)2]=1.2��10-11mol3•L-3��Ksp��AgCl��=1.8��10-10mol2•L-2
Ksp��Ag2CrO4��=1.9��10-12mol3•L-3��Ksp��CH3COOAg��=2.3��10-3mol2•L-2 ����������ȷ��
�� ��
A����������Ũ�Ⱦ�Ϊ0.2mol/L��AgNO3��Һ��CH3COONa��Һһ������CH3COOAg����
B����0.001mol/L��AgNO3��Һ����0.001mol/L��KCl��0.001mol/L��K2CrO4��Һ���Ȳ���Ag2CrO4����
C����Mg2+Ϊ0.121mol/L����Һ��Ҫ����Mg(OH)2��������Һ��pH����Ҫ������9����
D����AgClˮ��Һ�м���NaCl��Һ��Ksp(AgCl)���
AC

��ϰ��ϵ�д�
�����Ŀ
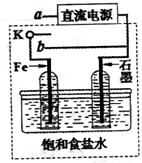 ��2013?�Ͳ���ģ����ˮ�Ƿḻ����Դ���⣬�Ӻ�ˮ����ȡԪ���ǻ�ѧ��ҵ����Ҫ��ɲ��֣�
��2013?�Ͳ���ģ����ˮ�Ƿḻ����Դ���⣬�Ӻ�ˮ����ȡԪ���ǻ�ѧ��ҵ����Ҫ��ɲ��֣�