��Ŀ����
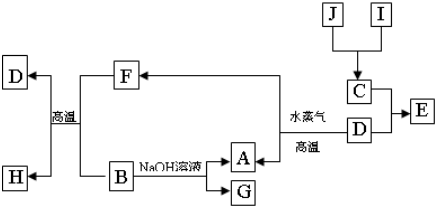
A��B��C��D�����ֳ����ĵ��ʣ�A��BΪ������C��D�����������壬��DΪ����ɫ���塣�ס��ҡ���Ϊ�����Ļ�������Ǻ�ɫ�Ҿ��д��Ե����ʡ�����֮���ת����ϵ����ͼ��ʾ��

��ش��������⣺
��1��B���Ӧ�Ļ�ѧ����ʽ��__________ ��
��2�������£���A��B�ĵ��ʷ���Ũ�����Ũ�����У��Ƿ��ܽ⣿________ ����ǡ�����
��3����������ˮ�����Һ��������������ӵķ�����____________ ��
��4��д��A��ˮ������Ӧ����C�ͼĻ�ѧ����ʽ_____________ ��
��5����A��B���ֽ�����һ������������ɻ���
��ȡһ�������ĸû��������м���������NaOH��Һ���������������ڱ�״����Ϊn L��B��
NaOH��Һ��Ӧ�����ӷ���ʽ��____________ ���������B�����ʵ���Ϊ_________ mol���ú���ĸ�ķ���ʽ��ʾ����
����ȡ��ͬ�����ĸû��������м���������ϡ���ᣬ����ȫ���ܽ⣬�������������ڱ�״����Ϊm L���÷�Ӧ��ת�Ƶ��ӵ����ʵ���Ϊ_________ mol���������A������Ϊ_________ g���ú���ĸ�ķ���ʽ��ʾ����
��������õ���Һ�м������������������Һ����ֽ��裬�������ij������˳�������ϴ�ӡ�������պ�õ�һ�ֹ��壬���������ָù����������ԭ����������ǡ����ȣ���ԭ�������������������Ϊ__________ ��
��1��B���Ӧ�Ļ�ѧ����ʽ��__________ ��
��2�������£���A��B�ĵ��ʷ���Ũ�����Ũ�����У��Ƿ��ܽ⣿________ ����ǡ�����
��3����������ˮ�����Һ��������������ӵķ�����____________ ��
��4��д��A��ˮ������Ӧ����C�ͼĻ�ѧ����ʽ_____________ ��
��5����A��B���ֽ�����һ������������ɻ���
��ȡһ�������ĸû��������м���������NaOH��Һ���������������ڱ�״����Ϊn L��B��
NaOH��Һ��Ӧ�����ӷ���ʽ��____________ ���������B�����ʵ���Ϊ_________ mol���ú���ĸ�ķ���ʽ��ʾ����
����ȡ��ͬ�����ĸû��������м���������ϡ���ᣬ����ȫ���ܽ⣬�������������ڱ�״����Ϊm L���÷�Ӧ��ת�Ƶ��ӵ����ʵ���Ϊ_________ mol���������A������Ϊ_________ g���ú���ĸ�ķ���ʽ��ʾ����
��������õ���Һ�м������������������Һ����ֽ��裬�������ij������˳�������ϴ�ӡ�������պ�õ�һ�ֹ��壬���������ָù����������ԭ����������ǡ����ȣ���ԭ�������������������Ϊ__________ ��
��1��3Fe3O4 + 8Al 9Fe + 4Al2O3
9Fe + 4Al2O3
��2����
��3��ȡ����������Һ���Թ��У��μ�KSCN��Һ������Һ��죬˵�����д���Fe3+
��4��3Fe + 4H2O (g) Fe3O4 + 4H2
Fe3O4 + 4H2
��5����2Al+2OH-+2H2O��2AlO2-+3H2�� �� ��
��
�� ��
�� ��
��
�� 30%
 9Fe + 4Al2O3
9Fe + 4Al2O3 ��2����
��3��ȡ����������Һ���Թ��У��μ�KSCN��Һ������Һ��죬˵�����д���Fe3+
��4��3Fe + 4H2O (g)
 Fe3O4 + 4H2
Fe3O4 + 4H2 ��5����2Al+2OH-+2H2O��2AlO2-+3H2�� ��
 ��
����
 ��
�� ��
���� 30%

��ϰ��ϵ�д�
�����Ŀ
a��b��c��d�����ֶ�����Ԫ�أ�a��b��dͬ���ڣ�c��dͬ���壮a��ԭ�ӽṹʾ��ͼΪ ��b��c�γɻ�����ĵ���ʽΪ
��b��c�γɻ�����ĵ���ʽΪ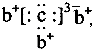 ���бȽ�����ȷ���ǣ�������
���бȽ�����ȷ���ǣ�������
 ��b��c�γɻ�����ĵ���ʽΪ
��b��c�γɻ�����ĵ���ʽΪ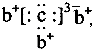 ���бȽ�����ȷ���ǣ�������
���бȽ�����ȷ���ǣ�������| A��ԭ�Ӱ뾶��a��c��d��b | B����ۺ����������c��d��a | C��ԭ��������a��d��b��c | D�����ʵ�������a��b��d��c |
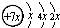 ��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺
��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺
 Al��OH��3+OH-
Al��OH��3+OH-