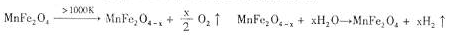��Ŀ����
��15�֣�
������Դ����Ľ�һ��ͻ���������Ȼ�ѧѭ��������о��ܵ��������ҵ�����������о����֣����������������̣�MnFe2O4���������Ȼ�ѧѭ���ֽ�ˮ���⡣
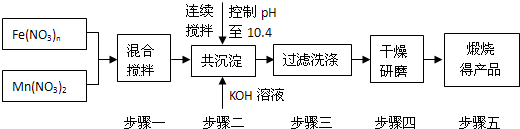
��MnFe2O4���Ʊ���

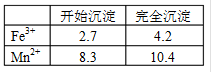
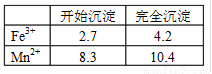
��֪Fe3+��Mn2+������pHΪ�±���ʾ��
��1���˹�����������Ͷ��ԭ��Fe(NO3)3��Mn(NO3)2
�����ʵ���֮��ӦΪ ��
��2���������b��ֵΪ ��
��3����������ϴ�Ӹɾ��ı��� ��
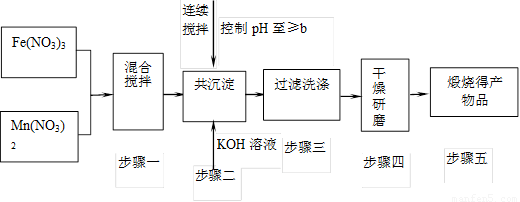
����MnFe2O4�Ȼ�ѧѭ�����⣺
MnFe2O4(s)��MnFe2O(4��x)(s) + ![]() O2(g)����H1
O2(g)����H1
MnFe2O(4��x)(s)+ xH2O ��MnFe2O4(s) + xH2(g)����H2
��ش��������⣺
��4�� ��MnFe2O(4��x)��x��0.8����MnFe2O(4��x)��Fe2+��ȫ����Ԫ���е���������Ϊ ��
��5�����Ȼ�ѧѭ��������ŵ��� ������ţ���
A�����̼�����Ⱦ B�����Ͽ�ѭ��ʹ�� C�������������ڲ�ͬ�������ɣ���ȫ������
��6����֪ 2H2��g��+O2��g���� 2H2O��g������H3
��H3����H1����H2�Ĺ�ϵΪ��H3= ��
��15��
��1��2��1 ��2��10.4 ��3������ϴ��Һ������ ����2�֣�
��4��80% ��5��A��B��C ��6����![]() ����H1 +��H2������3�֣�
����H1 +��H2������3�֣�
����:

 ��У����ϵ�д�
��У����ϵ�д�


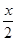 O2(g)����H1
O2(g)����H1