��Ŀ����
���ǵ����Ϻ����ḻ��һ��Ԫ�أ��䵥�ʼ��������ڹ�ũҵ������������������Ҫ���á�
��1��һ���¶��£���1L�ݻ��㶨���ܱ������г���2 mol N2��8molH2��������Ӧ��10min��ƽ�⣬��ð�����Ũ��Ϊ0��4 mol��L��1����ʱ������ת����Ϊ________��������߰����IJ��ʣ����ݻ�ѧƽ���ƶ�ԭ������������Ľ���______________��д��һ�����ɣ���
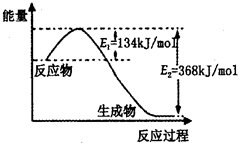
��2����ͼ��1mol NO2��g����1mol CO��g����Ӧ����lmol CO2��g����1 mol NO��g�������������仯ʾ��ͼ����д���÷�Ӧ���Ȼ�ѧ����ʽ_____________________��
��3�����ݻ��㶨���ܱ������У��������·�Ӧ��N2��g����3H2��g�� 2NH3��g����H��0����ƽ�ⳣ��K���¶�T�Ĺ�ϵ���±���
2NH3��g����H��0����ƽ�ⳣ��K���¶�T�Ĺ�ϵ���±���
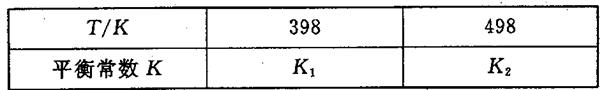
�ٸ÷�Ӧ��ƽ�ⳣ������ʽ��K��_____________��
�����ж�K1__________K2����д��������������������
��NH3��g��ȼ�յķ���ʽΪ��4NH3��g����7O2��g����4NO2��g����6H2O��l������֪��
��2H2��g����O2��g��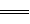 2H2O��l�� ��H����483��6 kJ��mol
2H2O��l�� ��H����483��6 kJ��mol
��N2��g����2O2��g��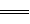 2NO2��g�� ��H����67��8 kJ��mol
2NO2��g�� ��H����67��8 kJ��mol
��N2��g����3H2��g��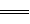 2NH3��g�� ��H����92��0 kJ��mol
2NH3��g�� ��H����92��0 kJ��mol
�����NH3��g����ȼ����________kJ��mol��
��1��һ���¶��£���1L�ݻ��㶨���ܱ������г���2 mol N2��8molH2��������Ӧ��10min��ƽ�⣬��ð�����Ũ��Ϊ0��4 mol��L��1����ʱ������ת����Ϊ________��������߰����IJ��ʣ����ݻ�ѧƽ���ƶ�ԭ������������Ľ���______________��д��һ�����ɣ���
��2����ͼ��1mol NO2��g����1mol CO��g����Ӧ����lmol CO2��g����1 mol NO��g�������������仯ʾ��ͼ����д���÷�Ӧ���Ȼ�ѧ����ʽ_____________________��

��3�����ݻ��㶨���ܱ������У��������·�Ӧ��N2��g����3H2��g��
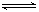 2NH3��g����H��0����ƽ�ⳣ��K���¶�T�Ĺ�ϵ���±���
2NH3��g����H��0����ƽ�ⳣ��K���¶�T�Ĺ�ϵ���±���
�ٸ÷�Ӧ��ƽ�ⳣ������ʽ��K��_____________��
�����ж�K1__________K2����д��������������������
��NH3��g��ȼ�յķ���ʽΪ��4NH3��g����7O2��g����4NO2��g����6H2O��l������֪��
��2H2��g����O2��g��
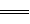 2H2O��l�� ��H����483��6 kJ��mol
2H2O��l�� ��H����483��6 kJ��mol��N2��g����2O2��g��
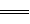 2NO2��g�� ��H����67��8 kJ��mol
2NO2��g�� ��H����67��8 kJ��mol��N2��g����3H2��g��
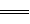 2NH3��g�� ��H����92��0 kJ��mol
2NH3��g�� ��H����92��0 kJ��mol�����NH3��g����ȼ����________kJ��mol��
��1��10%������Ӧ���Ũ�ȡ����¡�����ѹǿ�ȣ�
��2��NO2��g��+CO��g��=CO2��g��+NO��g����H=-234 kJ��mol��
��3����K=c2(NH3)/ c(N2) c3(H2)���ڣ�����282.8
��2��NO2��g��+CO��g��=CO2��g��+NO��g����H=-234 kJ��mol��
��3����K=c2(NH3)/ c(N2) c3(H2)���ڣ�����282.8
�����������1����������֪��ƽ��ʱ���������ʵ���Ϊ0.4mol������N2��g����3H2��g��
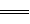 2NH3��g��֪��ת���ĵ��������ʵ���Ϊ0.2mol��������ת����Ϊ10%��������߰����IJ��ʣ����ݻ�ѧƽ���ƶ�ԭ������������Ľ��飺����Ӧ���Ũ�ȡ����¡�����ѹǿ�ȣ���2�������ͼ��֪���÷�Ӧ���ʱ��H=E1��E2=134KJ/mol��368KJ/mol=��234KJ/mol���Ȼ�ѧ����ʽΪNO2��g��+CO��g��=CO2��g��+NO��g����H=��234 kJ��mol����3���ٸ��ݻ�ѧƽ�ⳣ���Ķ���д����K=c2(NH3)/ c(N2) c3(H2)���ںϳɰ��ķ�ӦΪ���ȷ�Ӧ�������¶ȣ�ƽ�������ƶ���ƽ�ⳣ����С������K1��K2���۸��ݸ�˹���ɣ��١�3+�ڡ�2���ۡ�2�ã�4NH3��g����7O2��g����4NO2��g����6H2O��l������H=��1131.2 kJ��mol�����ȼ���ȵĶ���֪��NH3��g����ȼ����282.8kJ��mol��
2NH3��g��֪��ת���ĵ��������ʵ���Ϊ0.2mol��������ת����Ϊ10%��������߰����IJ��ʣ����ݻ�ѧƽ���ƶ�ԭ������������Ľ��飺����Ӧ���Ũ�ȡ����¡�����ѹǿ�ȣ���2�������ͼ��֪���÷�Ӧ���ʱ��H=E1��E2=134KJ/mol��368KJ/mol=��234KJ/mol���Ȼ�ѧ����ʽΪNO2��g��+CO��g��=CO2��g��+NO��g����H=��234 kJ��mol����3���ٸ��ݻ�ѧƽ�ⳣ���Ķ���д����K=c2(NH3)/ c(N2) c3(H2)���ںϳɰ��ķ�ӦΪ���ȷ�Ӧ�������¶ȣ�ƽ�������ƶ���ƽ�ⳣ����С������K1��K2���۸��ݸ�˹���ɣ��١�3+�ڡ�2���ۡ�2�ã�4NH3��g����7O2��g����4NO2��g����6H2O��l������H=��1131.2 kJ��mol�����ȼ���ȵĶ���֪��NH3��g����ȼ����282.8kJ��mol��
��ϰ��ϵ�д�
�����Ŀ

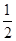 O2(g)=H2O(g) ��H1����241.8 kJ��mol��1
O2(g)=H2O(g) ��H1����241.8 kJ��mol��1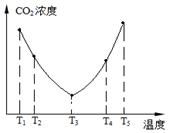
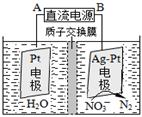
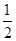 O2(g) ===CO(g)����H1 C(s)+O2(g) ===CO2(g)����H2
O2(g) ===CO(g)����H1 C(s)+O2(g) ===CO2(g)����H2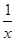 Sx��s�� ��H��akJ/mol
Sx��s�� ��H��akJ/mol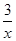 Sx��s�� ��H��bkJ/mol��
Sx��s�� ��H��bkJ/mol��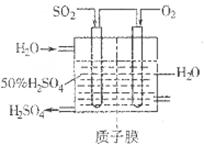
 2SO3��g�� ��H��0��SO2��ת�����ǿ���SO2�ŷŵĹؼ���ʩ֮һ��ij����С�����������̽����
2SO3��g�� ��H��0��SO2��ת�����ǿ���SO2�ŷŵĹؼ���ʩ֮һ��ij����С�����������̽���� 2SO3��g����Ӧ��Ӱ�죬ʵ������ͼ��ʾ����ͼ��T��ʾ�¶ȣ�n��ʾ���ʵ���������a��b��c����������ƽ��״̬�У�SO2��ת������ߵ���____���¶�T1______T2���������������������
2SO3��g����Ӧ��Ӱ�죬ʵ������ͼ��ʾ����ͼ��T��ʾ�¶ȣ�n��ʾ���ʵ���������a��b��c����������ƽ��״̬�У�SO2��ת������ߵ���____���¶�T1______T2���������������������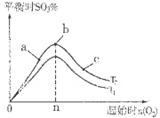
 2NH3 ��H ="-92.4" kJ/mol,��ʼ���ǽ�N2��H2�������20mol (�����1��1)����5L�ϳ�����.��ӦǰѹǿΪP0,��Ӧ������ѹǿ��P��ʾ����Ӧ������P/P0��ʱ��t�Ĺ�ϵ��ͼ��ʾ����ش��������⣺
2NH3 ��H ="-92.4" kJ/mol,��ʼ���ǽ�N2��H2�������20mol (�����1��1)����5L�ϳ�����.��ӦǰѹǿΪP0,��Ӧ������ѹǿ��P��ʾ����Ӧ������P/P0��ʱ��t�Ĺ�ϵ��ͼ��ʾ����ش��������⣺
 CH3OH(g) ��H
CH3OH(g) ��H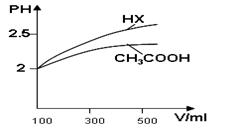
 CH3OH(g) ��H1
CH3OH(g) ��H1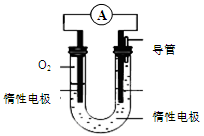
 N������941kJ��������1molN4����ת��Ϊ2molN2ʱҪ�ų�______________ kJ������
N������941kJ��������1molN4����ת��Ϊ2molN2ʱҪ�ų�______________ kJ������