��Ŀ����
A��B��C��D��EΪǰ20��Ԫ�أ�ԭ������������������A��Dͬ���壬B��C��Dͬ���ڣ�B��C��D����������Ӧ��ˮ����������Ӧ�����κ�ˮ��A��B��E�ĵ��ʾ��ܺ�ˮ���ҷ�Ӧ��E+��D�������Ӿ�����ͬ�ĵ��Ӳ�ṹ��
��1��A��B��C��D����Ԫ�ص�ԭ�Ӱ뾶��С�����˳���� ����Ԫ�ط��ű�ʾ����
��2��A��D���⻯���� ���û�ѧʽ��ʾ���ķе�ϸߣ�ԭ���� ��
��3��д��E������ˮ��Ӧ�����ӷ���ʽ ��
��4��B��D�γɵľ����У�ÿ����������������ҵȾ�������ӹ��� ����
��1��A��B��C��D����Ԫ�ص�ԭ�Ӱ뾶��С�����˳����
��2��A��D���⻯����
��3��д��E������ˮ��Ӧ�����ӷ���ʽ
��4��B��D�γɵľ����У�ÿ����������������ҵȾ�������ӹ���
������A��B��C��D��EΪǰ20��Ԫ�أ�ԭ��������������B��C��Dͬ���ڣ�B��C��D����������Ӧ��ˮ����������Ӧ�����κ�ˮ��Ӧ������������ǿ�ǿ��ķ�Ӧ����CΪAlԪ�ء�BΪNaԪ�أ�D����ΪS��Cl��E+��D�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����EΪKԪ�أ�A��Dͬ���壬��A�ĵ����ܺ�ˮ���ҷ�Ӧ����AΪFԪ�ء�DΪClԪ�أ��ݴ˽��
����⣺A��B��C��D��EΪǰ20��Ԫ�أ�ԭ��������������B��C��Dͬ���ڣ�B��C��D����������Ӧ��ˮ����������Ӧ�����κ�ˮ��Ӧ������������ǿ�ǿ��ķ�Ӧ����CΪAlԪ�ء�BΪNaԪ�أ�D����ΪS��Cl��E+��D�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����EΪKԪ�أ�A��Dͬ���壬��A�ĵ����ܺ�ˮ���ҷ�Ӧ����AΪFԪ�ء�DΪClԪ�أ�
��1��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����ԭ�Ӱ뾶F��Cl��Al��Na��
�ʴ�Ϊ��F��Cl��Al��Na��
��2������HF����֮������������HF�ķе����HCl��
�ʴ�Ϊ��HF��HF����֮����������
��3��K��ˮ��Ӧ����������������������Ӧ���ӷ���ʽΪ��2K+2H2O=2K++2OH-+H2����
�ʴ�Ϊ��2K+2H2O=2K++2OH-+H2����
��4��B��D�γ�NaCl���壬�����ṹ��ͼ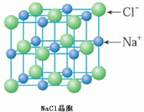 �������ĵ�������Ϊ�о�������֮����������Ӵ��ھ��������ϣ�ÿ����������Χ��12�������ӣ�
�������ĵ�������Ϊ�о�������֮����������Ӵ��ھ��������ϣ�ÿ����������Χ��12�������ӣ�
�ʴ�Ϊ��12��
��1��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����ԭ�Ӱ뾶F��Cl��Al��Na��
�ʴ�Ϊ��F��Cl��Al��Na��
��2������HF����֮������������HF�ķе����HCl��
�ʴ�Ϊ��HF��HF����֮����������
��3��K��ˮ��Ӧ����������������������Ӧ���ӷ���ʽΪ��2K+2H2O=2K++2OH-+H2����
�ʴ�Ϊ��2K+2H2O=2K++2OH-+H2����
��4��B��D�γ�NaCl���壬�����ṹ��ͼ
 �������ĵ�������Ϊ�о�������֮����������Ӵ��ھ��������ϣ�ÿ����������Χ��12�������ӣ�
�������ĵ�������Ϊ�о�������֮����������Ӵ��ھ��������ϣ�ÿ����������Χ��12�������ӣ��ʴ�Ϊ��12��
���������⿼��ṹλ�����ʹ�ϵӦ�ã��Ѷ��еȣ�ע�������������������Խ����ƶϣ���4��Ϊ�״��㣬ע��ʶ����ѧ���������ṹ��

��ϰ��ϵ�д�
 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д� Ӣ�żƻ���ĩ����ϵ�д�
Ӣ�żƻ���ĩ����ϵ�д�
�����Ŀ
����ѧ--ѡ��3�����ʽṹ�����ʡ�
��������Ԫ�أ�����A��B��C��D��EΪ����������Ԫ�أ�F��GΪ��������Ԫ�أ����ǵ�ԭ����������������������������Ϣ���ش����⣮
��1��Gλ�� �� �����۵����Ų�ʽΪ ��
��2��B��̬ԭ����������ߵĵ��ӣ���������ڿռ��� ������ԭ�ӹ����
�Σ�
��3������Cԭ�ӵĵ����Ų�ͼ ��
��4����֪BA5Ϊ���ӻ����д�������ʽ ��
��5��DE3����ԭ�ӵ��ӻ���ʽΪ ���ü۲���ӶԻ��������Ʋ���ռ乹��Ϊ ��
��6���õ���ʽ��ʾFԪ����EԪ���γɻ�������γɹ��� ��
��������Ԫ�أ�����A��B��C��D��EΪ����������Ԫ�أ�F��GΪ��������Ԫ�أ����ǵ�ԭ����������������������������Ϣ���ش����⣮
| AԪ�صĺ���������͵��Ӳ�����ȣ�Ҳ����������ḻ��Ԫ�� |
| BԪ��ԭ�ӵĺ���p��������s��������1 |
| Cԭ�ӵĵ�һ�����ĵ����ֱܷ��ǣ� I1=738kJ/mol I2=1451kJ/mol I3=7733kJ/mol I4=10540kJ/mol |
| Dԭ�Ӻ�������p���ȫ������� |
| EԪ�ص������������������IJ�Ϊ4 |
| F��ǰ�������е縺����С��Ԫ�� |
| G�����ڱ��ĵ����� |
��2��B��̬ԭ����������ߵĵ��ӣ���������ڿռ���
�Σ�
��3������Cԭ�ӵĵ����Ų�ͼ
��4����֪BA5Ϊ���ӻ����д�������ʽ
��5��DE3����ԭ�ӵ��ӻ���ʽΪ
��6���õ���ʽ��ʾFԪ����EԪ���γɻ�������γɹ���
 A��B��C��D��EΪ��ѧ��ѧ�������ʣ�����A��CΪ�������ʣ�EΪ�ǽ������ʣ���ͼ������֮����ת����ϵ����ش�
A��B��C��D��EΪ��ѧ��ѧ�������ʣ�����A��CΪ�������ʣ�EΪ�ǽ������ʣ���ͼ������֮����ת����ϵ����ش�

