��Ŀ����
�л�������G�Ǻϳ�ά������ҩ����м��壬��ṹ��ʽΪ��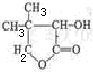
G�ĺϳ���·���£�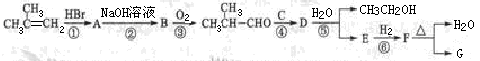
����A��F�ֱ����һ���л�������ϳ�·���еIJ��ֲ��P��Ӧ��������ȥ��
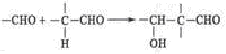
��֪��
��ش��������⣺
��1��G�ķ���ʽ�� �� ��G�й����ŵ������� �� ��
��2���ڢٲ���Ӧ�Ļ�ѧ����ʽ�� ��
��3��B�����ƣ�ϵͳ�������� ��
��4���ڢڡ���������ȡ����Ӧ���� ������ţ���
��5���ڢܲ���Ӧ�Ļ�ѧ����ʽ�� ��
��6��д��ͬʱ��������������E������ͬ���칹��Ľṹ��ʽ ��
��ֻ����һ�ֹ����ţ�����״�ṹ���ޡ�O��O�����ۺ˴Ź�������ֻ��2�ַ塣
��1����C6H10O3���������ǻ�
��2��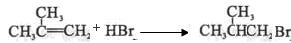
��3��2-��-1-�����Ȣڢ�
��5��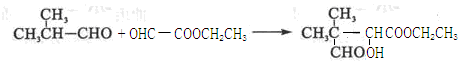
��6��CH3COOCH2CH2OOCCH3��CH3OOCCH2CH2COOCH3��CH3CH2OOCCOOCH2CH3
����

��ϰ��ϵ�д�
�����Ŀ
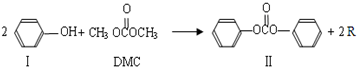
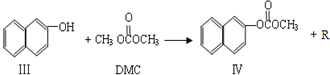
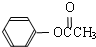 ���ڴ���������Ҳ�����ɻ������ͬʱ�õ�һ�ָ���ƷG�������й�G��˵����ȷ����
���ڴ���������Ҳ�����ɻ������ͬʱ�õ�һ�ָ���ƷG�������й�G��˵����ȷ����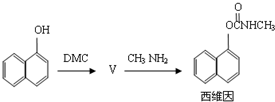



 ���ڴ���������Ҳ�����ɻ������ͬʱ�õ�һ�ָ���ƷG�������й�G��˵����ȷ����_______��
���ڴ���������Ҳ�����ɻ������ͬʱ�õ�һ�ָ���ƷG�������й�G��˵����ȷ����_______��