��Ŀ����
��7�֣���A��B��C��D��E���ֶ�����Ԫ�أ���ԭ��������������Aԭ�ӵĵ��Ӳ��������ĺ������������ͬ��Bԭ�Ӻ��������������Ǵ�����������2����CԪ�ص�ԭ�ӻ�̬ʱ2p�ܼ���2s�ܼ��ϵĵ�������2����D�Ǹ�������ԭ�Ӱ뾶��������Ԫ�أ�Eԭ�ӵĵ�һ�����ĵ����ܣ�kJ��mol��1���ֱ�Ϊ��578��1817��2745��11575����ش��������⣺
��1�� C��D��Ԫ���γɵĻ�����ľ��������������� ��BC2�Ľṹʽ������������
��2����F��D��ͬ������һ����Ԫ�أ���F�ĵ����Ų�ʽ�ǣ�����������������������
��3��д��E������������Ӧ��ˮ������D������������Ӧ��ˮ���ﷴӦ�����ӷ���ʽ���� �� ��
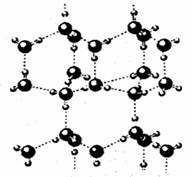
��4��A2C���Ӽ�����ڡ�����������ö��˴˽���γ�(A2C)n���ڸ÷����γɵľ�����ÿ��A2C���ӱ�4��A2C���Ӱ�Χ�γɱ��ε������壬ͨ�������������ӳ��Ӵ�ķ��Ӿ��壬��ṹʾ��ͼ����ͼ��ʾ������ա�
�ٺ�1molA2C�ĸþ�������_____mol���������
����G��C��ͬ������һ����Ԫ�أ���Ƚ�A2C��A2G�е�ĸߵ�______________��
(1)���Ӿ��� O=C=O ��2��1S22S22P63S23P64S1 ��3��Al(OH)3+OH-=AlO2-+2H2O
(4)�� 2 ��A2C > A2G (��H2O > H2S )
�������� ����Ԫ�صĽṹ�ص㼰���ʿ�֪��A��B��C��D��E���ֶ�����Ԫ�طֱ���H��C��O��Na��Al��
��1���ƺ����γɵ������ﺬ�����Ӽ��������Ӿ��塣CO2��ֱ���ͽṹ���ṹʽΪO=C=O��
��2��D���ƣ���F��K�����ݹ���ԭ����֪��������Ų�ʽΪ1S22S22P63S23P64S1��
��3�����������������������������ǿ���У�����ʽΪAl(OH)3+OH-=AlO2-+2H2O��
��4��ÿ��A2C���ӱ�4��A2C���Ӱ�Χ�γɱ��ε������壬����ƽ��ÿ��ˮ�����γ�2�����������ˮ�����д��������������е��������ġ�






