��Ŀ����
ʵ������ȡ
Cl2(1)ԭ��
��ѧ����ʽΪ__________��
���ӷ���ʽΪ__________��
��Ӧ��________����������________�ǻ�ԭ����
(2)װ��

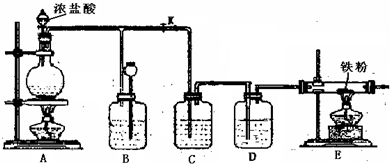
��A���������巢��װ�ã�����������Ϊ________��________��________(��________)��________��________��________��________��
��B����������________װ�ã�
��C������________װ�ã�
(3)�ռ�������________����________��
(4)��������
����Cl2�ǻ���ɫ���壬���۲쵽________����֤�����ռ���Cl2��
�ڿ��ڼ���ƿ�ڷ�һʪ��ĵ��۵⻯����ֽ����________����˵�����ռ���Cl2��
(5)Cl2�ľ�������Cl2��ͨ��ʢ________��ϴ��ƿ����ͨ��ʢ________��ϴ��ƿ��Ȼ����________���ռ�����ɵõ������Ҹ����Cl2��
(6)β����������________���ն����Cl2����ѧ����ʽΪ________�����ӷ���ʽΪ________��
�𰸣�

��ϰ��ϵ�д�
�����Ŀ