��Ŀ����
��һ���ܱ������н������·�Ӧ��2X2��g����Y2��g��  2Z��g������Ӧ������ijһʱ��X2��Y2��Z��Ũ�ȷֱ�Ϊ0.2 mol��L��1��0.1 mol��L��1��0.2 mol��L��1������Ӧ�ﵽƽ��ʱ���������ݿ��ܺ������ǣ� ��
2Z��g������Ӧ������ijһʱ��X2��Y2��Z��Ũ�ȷֱ�Ϊ0.2 mol��L��1��0.1 mol��L��1��0.2 mol��L��1������Ӧ�ﵽƽ��ʱ���������ݿ��ܺ������ǣ� ��
A��Z��Ũ��Ϊ0.4 mol��L��1 B��X2��Z��Ũ�Ⱦ�Ϊ0.15 mol��L��1
C��X2��Ũ��Ϊ0.4 mol��L��1 D��X2��Z��Ũ��֮��Ϊ0.4 mol��L��1
��ϰ��ϵ�д�
�����Ŀ


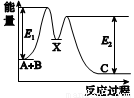
 2CO(g)��Ӧ�У���ʹ��Ӧ��������Ĵ�ʩ��
2CO(g)��Ӧ�У���ʹ��Ӧ��������Ĵ�ʩ�� NaCl+NH3��+H2O
NaCl+NH3��+H2O ��ʾ��
��ʾ��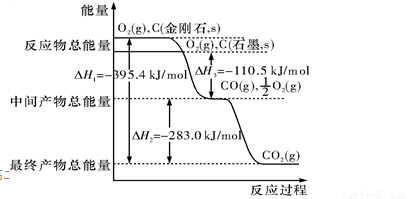
 ��ȼ�գ������������ֹ�̬������P2O3��P2O5 �� P2O5��ͨ��״̬���ȶ�����֪3.1 g�ĵ����ף�P����3.2 g������ȼ�գ�����Ӧ��ľ����ų�X kJ������
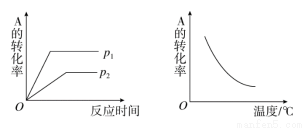
��ȼ�գ������������ֹ�̬������P2O3��P2O5 �� P2O5��ͨ��״̬���ȶ�����֪3.1 g�ĵ����ף�P����3.2 g������ȼ�գ�����Ӧ��ľ����ų�X kJ������ xC��g���ڲ�ͬ�¶ȼ�ѹǿ��p1��p2�������·�Ӧ
xC��g���ڲ�ͬ�¶ȼ�ѹǿ��p1��p2�������·�Ӧ ��A��ת���ʵı仯�����ͼ��ʾ�������ж���ȷ���ǣ� ��
��A��ת���ʵı仯�����ͼ��ʾ�������ж���ȷ���ǣ� ��