��Ŀ����
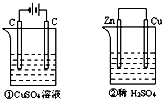 ��ͼ��ʾ������װ���У���Һ�������Ϊ200mL����ʼʱ�������Һ��Ũ�Ⱦ�Ϊ0.1mol/L������һ��ʱ���õ�����ͨ��0.02mol���ӣ����������ε�ˮ�����Һ������仯��������������ȷ���ǣ�������
��ͼ��ʾ������װ���У���Һ�������Ϊ200mL����ʼʱ�������Һ��Ũ�Ⱦ�Ϊ0.1mol/L������һ��ʱ���õ�����ͨ��0.02mol���ӣ����������ε�ˮ�����Һ������仯��������������ȷ���ǣ�������������������ӵ�Դ���ǵ��أ���û����ӵ�Դ����ԭ��أ�����������4OH--4e-=2H2O+O2������������Cu2++2e-=Cu������п�Ļ����Դ���ͭ�ģ�����п�Ǹ�����ͭ��������������ӦΪ2H++2e-=H2������������Zn-2e-=Zn2+���Դ˽����⣮
����⣺������ͨ��0.02mol���ӣ�
A������������4OH--4e-=2H2O+O2��������0.005molO2����������ӦΪ2H++2e-=H2��������0.01molH2����������������٣��ڣ���A����
B��������OH-������H+����ҺpH��С��������H+����ҺpH����B����
C������������ͭ������������������û�й�����������缫�������������ʵ������٣��ڣ���C��ȷ��
D�����и�����ӦΪZn-2e-=Zn2+����D����
��ѡC��
A������������4OH--4e-=2H2O+O2��������0.005molO2����������ӦΪ2H++2e-=H2��������0.01molH2����������������٣��ڣ���A����
B��������OH-������H+����ҺpH��С��������H+����ҺpH����B����
C������������ͭ������������������û�й�����������缫�������������ʵ������٣��ڣ���C��ȷ��
D�����и�����ӦΪZn-2e-=Zn2+����D����
��ѡC��
���������⿼��ԭ��غ͵��صĹ���ԭ����ע����յ缫����ʽ����д��Ϊ������Ĺؼ���ע��ԭ��غ͵��ص�������Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
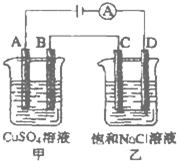 �ش����и��⣺
�ش����и��⣺
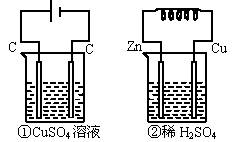
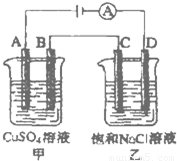 �ش����и��⣺
�ش����и��⣺