��Ŀ����
��ʾ���б仯��ʽ����ȷ���ǣ� ��
A��NaHCO3ˮ������ӷ���ʽ��HCO![]() +H2O
+H2O ![]() H3O++CO
H3O++CO![]()
B��ʯ��ʯ���ڴ�������ӷ���ʽ��CaCO3+2H+=Ca2++CO2��+H2O
C�������绯ѧ��ʴ�ĸ�����Ӧ��4OH����4e��=2H2O+O2��
D��1 L 0.5mol/L H2SO4��Һ��1 L 1.0mol/L NaOH��Һ��Ӧ���ų�57.3kJ��������
1/2H2SO4(aq)+NaOH(aq)=1/2Na2SO4(aq)+H2O(l)����H=��57.3kJ/mol
D

��ϰ��ϵ�д�
�����Ŀ
 N2(g)+2CO2(g)
N2(g)+2CO2(g) N2(g) +2CO2(g)�ġ�H=___________��
N2(g) +2CO2(g)�ġ�H=___________��


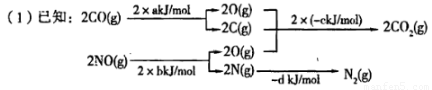

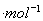

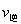
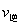
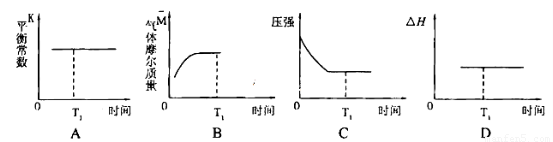
 N2(g)+2CO2(g)
N2(g)+2CO2(g) N2(g) +2CO2(g)�ġ�H=___________��
N2(g) +2CO2(g)�ġ�H=___________��
