��Ŀ����
16����ͼ��Na��Cu��H��C��N��Ԫ�ص��ʵ��۵�ߵ͵�˳������c��d�����Ⱥ͵�������壮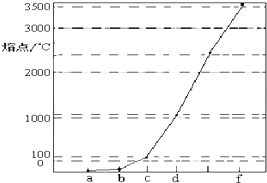
��1����д����ͼ��d���ʶ�ӦԪ��ԭ�ӵĵ����Ų�ʽ1s22s22p63s23p63d104s1
��2������a��b��f��Ӧ��Ԫ����ԭ�Ӹ�����1��1��1�γɵķ����к�2���Ҽ�����2���м���
��3��a��b��Ԫ���γɵ�10�������Է���X�Ŀռ乹��Ϊ�����Σ���X����ˮ�����Һ���뵽��dԪ�ظ����ӵ���Һ�������������ɵĺ�dԪ�����ӵĻ�ѧʽΪCu��NH3��4]2+������X��d�ĸ�����֮������λ������ϣ�
��4����������������Ԫ���е�һ��Ԫ���γɵĺ�����Ľṹ��
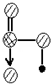 ���Ҫ˵��������������ˮ��ԭ��
���Ҫ˵��������������ˮ��ԭ����HNO3�Ǽ��Է��ӣ������ڼ��Ե�ˮ��
��HNO3�����е�-OH����ˮ����֮���γ������
���� �ڸ�����Ԫ���У�c��d�����Ⱥ͵�������壬����Ϊ������ͼ��d���۵��c�ĸߣ���֪dΪCu��cΪNa���ɵ��ʵ��۵�ߵ͵�˳���֪aΪ��������bΪ������fΪ̼���ʣ�
��1��dΪCu��ԭ�Ӻ��������Ϊ29�����ݺ�������Ų�����д��Cu�ĵ����Ų�ʽ��
��2������a��b��f��Ӧ��Ԫ����ԭ�Ӹ�����1��1��1�γɵķ���ΪHCN���ṹʽΪH-C��N��
��3��aΪ������bΪ��������ӦԪ���γɵ�10�������Է���XΪ����������������ˮ�����Һ���뵽��Cu2+����Һ��������������[Cu��NH3��4]2+��
��4�����ݺ�����Ľṹ��֪����ΪHNO3���ٸ�����������ԭ������ˮ����֮���γ�����������
��� �⣺�ڸ�����Ԫ���У�c��d�����Ⱥ͵�������壬����Ϊ������ͼ��d���۵��c�ĸߣ���֪dΪCu��cΪNa���ɵ��ʵ��۵�ߵ͵�˳���֪aΪ��������bΪ������fΪ̼���ʣ�
��1��dΪCu��ԭ�Ӻ��������Ϊ29��ԭ�ӵĵ����Ų�ʽΪ1s22s22p63s23p63d104s1��
�ʴ�Ϊ��1s22s22p63s23p63d104s1��
��2������a��b��f��Ӧ��Ԫ����ԭ�Ӹ�����1��1��1�γɵķ���ΪHCN���ṹʽΪH-C��N�������к� 2���Ҽ�����2���м���
�ʴ�Ϊ��2��2��
��3��aΪ������bΪ��������ӦԪ���γɵ�10�������Է���XΪNH3���ռ乹��Ϊ�����ͣ�����������ˮ�����Һ���뵽��Cu2+����Һ��������������[Cu��NH3��4]2+��Cu2+������NH3�����γ���λ����
�ʴ�Ϊ�������Σ�Cu��NH3��4]2+�����ۼ�����λ����
��4�����ݺ�����Ľṹ��֪����ΪHNO3��HNO3�Ǽ��Է��ӣ������ڼ��Ե�ˮ�У�����HNO3�����е�-OH����ˮ����֮���γ������������ˮ��
�ʴ�Ϊ��HNO3�Ǽ��Է��ӣ������ڼ��Ե�ˮ�У�HNO3�����е�-OH����ˮ����֮���γ����
���� ���⿼���Ƕ����ʽṹ�����ʵĿ��飬�漰�������������ʡ���������Ų�����ѧ�����������ӽṹ�����ʵȣ��Ѷ��еȣ�ּ�ڿ����֪ʶ���ۺ����ú����գ�

 ����ѧУ�ֲ����ܲ�ϵ�д�
����ѧУ�ֲ����ܲ�ϵ�д� �ƸԺ���ȫ�����Ų��Ծ�ϵ�д�
�ƸԺ���ȫ�����Ų��Ծ�ϵ�д���1������д����Ŀհ״���
| ����� | ������ | ����� | |||
| ���ڸ�������� | �� | ��� | �� |
��3������ᷢ����Ӧ�����ӷ���ʽΪCaO+2H+=Ca2++H2O��
��4���ߢ������Լ����Բ��ö��ַ���������д������һ�ַ����ù������䣬�ܹ����������ЧӦ���ǵ�����Һ������Ϊ��������Һ��
a��������Ӧ��b��ȡ����Ӧ��c����ȥ��Ӧ��d���ӳɷ�Ӧ��e��ˮ�ⷴӦ�����������л�������������ǻ������ŵķ�Ӧ��ȷ����ǣ�������
| A�� | a��b��c | B�� | d��e | C�� | b��d��e | D�� | b��c��d��e |
| A�� | �ö����ЧӦ�����������������ϡ���� | |
| B�� | �ù��˵ķ�����ȥNaCl��Һ�к��е��������۽��� | |
| C�� | ���ܽ⡢���˵ķ����ᴿ��������BaSO4��BaCO3 | |
| D�� | �ü��ȷ������Գ�ȥMgCl2�е�NH4HCO3 |


 ��д������ԭ�Ӱ뾶��С��ԭ���γ�10������Ϊ��������ṹ�Ļ�����ĵ���ʽ
��д������ԭ�Ӱ뾶��С��ԭ���γ�10������Ϊ��������ṹ�Ļ�����ĵ���ʽ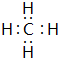 ���õ���ʽ��ʾ�ݺ͢��γɻ�����Ĺ���
���õ���ʽ��ʾ�ݺ͢��γɻ�����Ĺ��� ��
��