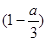��Ŀ����
ij�Ͻ�(����ͭ����)��ͭ���������ʵ���֮��Ϊymol������Cu�����ʵ�������Ϊa ������ȫ��Ͷ��50mLbmol��L��1��������Һ�У�����ʹ���ַ�Ӧ(����NO��Ψһ�Ļ�ԭ����)������˵������ȷ���ǣ� ��
A����������ʣ�࣬����Һ���ٵ�����������ֿ�ʼ�ܽ�
B��������ȫ���ܽ⣬����Һ��һ������Fe3��
C.��80y(1��a/3)��b��160y/3�������������ǡ����ȫ��Ӧ
D��������ȫ���ܽ⣬�Ҳ���336mL����(��״��)����b=0.3
BD
����:
A��,�μ������,��Һ�е�NO3-������������,������ʣ��Ľ���.B�У�������ȫ���ܽ�ʱ����Ԫ�ؿ�����Fe2+����ʽ���ڣ���һ����Fe3+��B����ȷ��C�У����ݷ�Ӧʽ��3Cu+8HNO3=Cu(NO3)2 +2NO��+4H2O ��3Fe+8HNO3=Fe(NO3)2+2NO��+4H2O ��Fe+4HNO3=Fe(NO3)3+NO��+2H2O �����ü�����������������ǡ����ȫ��Ӧʱ��80y(1��a/3)��b��160y/3 ��C��ȷ��D�У���b=0.3ʱ���μӷ�Ӧ������Ϊ0.05��0.3=0.015mol�������ɵ�NO�����ʵ���Ϊ![]() ,�μӷ�Ӧ����������ʵ���Ӧ�ô�������NO�����ʵ�����D����ȷ��
,�μӷ�Ӧ����������ʵ���Ӧ�ô�������NO�����ʵ�����D����ȷ��

 )
)