��Ŀ����
10���������л�ѧ��������Ʒ���ٲ����� ���ձ� ��������ƽ ����Ͳ ��ҩ�� ��ͷ�ι� ������ƿ ���ǩֽ ��ϸ���Լ�ƿ����������Ҫ����500mL 0.622moL/L NaOH��Һ����Ӧ��ȡ����NaOH12.4g��
�������ϳ������NaOH����������Һ�����ã���������Һ�Ĺ���˳������������������Ʒ��ѡȡ��Ҫ���������ظ��IJ��ƣ������������Ǣڢܢ٢ߢޢ�ࣻ
�����ƹ����У�Ӧ������NaOH�ܽ����ȴ�����µ���Һת�Ƶ�500mL����ƿ�У���ת��ʱ���������������ã���Һ��ӽ�����ƿ�̶���1-2cm�������ý�ͷ�ιܶ��ݣ�
������ʵ��������������������ܵ�����ҺŨ��ƫ�ߵ���AE������ĸ����
A���������������ܽ�ʱδ��ȴ�����£��Ϳ�ʼת�ơ�ϴ���Լ����ݣ�
B��ת��ʱ��С����������Һ����������ƿ�⣻
C������ʱ��������ƿ�̶��ߣ�
D������ʱ��ˮ�����˿̶��ߣ������ý�ͷ�ι���ȥ�����ˮ��
E�����ƺú��ֳ���ʱ���õ�������ƽ�����������ˣ�
���� I����������m=CVM�����㣻
II���������Ʋ����Ǽ��㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��������Ҫ��������
III������ƿ�������ȣ�����Һʱ�������������������������ݶ��ݵIJ�����������
IV������c=$\frac{n}{V}$��������ʵ����ʵ���n����Һ�����V�ı仯��������������
��� �⣺I�������������Ƶ�����m=CVM=0.622mol/L��0.5L��40g/mol=12.4g���ʴ�Ϊ��12.4��
II���������Ʋ����Ǽ��㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��֪�����������˳��Ϊ�ڢܢ٢ߢޢ�࣬�ʴ�Ϊ���ڢܢ٢ߢޢ�ࣻ
III������ƿ�������ȣ���������������ˮ���ȣ���Ҫ��������ȴ������Ȼ��ת����500mL����ƿ�У�����Һʱ������������������������ֹ��Һ����������ʱ��Ҫ��������ƿ�е�ˮ����Һ����̶���1-2cmʱ�����ý�ͷ�ι���μ��룬����Һ����̶������У�
�ʴ�Ϊ��500ml����ƿ�� ������ 1-2cm�� ��ͷ�ιܣ�
IV��A���������������ܽ�ʱδ��ȴ�����£��Ϳ�ʼת�ơ�ϴ���Լ����ݣ�����ȴ����Һ���ƫС��Ũ��ƫ�ߣ���A��ȷ��
B��ת��ʱ��С����������Һ����������ƿ�⣬��ᵼ�����ʵ���ʧ��Ũ��ƫС����B����
C������ʱ��������ƿ�̶��ߣ�����Һ���ƫ��Ũ��ƫ�ͣ���C����
D������ʱ��ˮ�����˿̶��ߣ������ý�ͷ�ι���ȥ�����ˮ���������IJ�ֹ��ˮ���������ʣ���Ũ��ƫ�ͣ���D����
E�����ƺú��ֳ���ʱ���õ�������ƽ�����������ˣ�������������������������ʵ���������Ũ��ƫ�ߣ���E��ȷ��
��ѡAE��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƹ����еļ���������������ڻ�������Ŀ���ѶȲ���

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
��1��ͼ����1mol NO2��g����1mol CO��g����Ӧ����1mol CO2��g����1mol NO��g�������е������仯ʾ��ͼ�����ڷ�Ӧ��ϵ�м����������Ӧ��������E1�ı仯�Ǽ�С�����������С�����䡱����ͬ������H�ı仯�Dz��䣮��д��NO2��CO��Ӧ���Ȼ�ѧ����ʽ��NO2 ��g��+CO ��g���TCO2 ��g��+NO ��g����H=-234 kJ•mol-1��
��2���״����ӽ���Ĥȼ�ϵ���н��״�����ת��Ϊ���������ַ�Ӧ�Ȼ�ѧ����ʽ���£�
��CH3OH��g��+H2O��g���TCO2��g��+3H2��g����H=+49.0kJ•mol-1
��CH3OH��g��+$\frac{1}{2}$O2��g���TCO2��g��+2H2��g����H=-192.9kJ•mol-1
��֪��H2O��g���TH2O��l����H=-44kJ•mol-1����״�����ȼ��ΪҺ̬ˮ���Ȼ�ѧ����ʽΪCH3OH ��g��+$\frac{3}{2}$O2 ��g���TCO2 ��g��+2 H2O ��l����H=-764.7 kJ•mol-1��
��3�������ʾ�Dz��ֻ�ѧ���ļ��ܲ�����
| ��ѧ�� | P-P | P-O | O�TO | P�TO |
| ����/kJ•mol-1 | a | b | c | x |
| A�� | �����ص�һ�������еĺ���42��ԭ�� | |
| B�� | ��������̼Ԫ�ص���������ԼΪ63.8% | |
| C�� | �����ص���Է�������Ϊ282 | |
| D�� | 0.1mol�����ص�����Ϊ28.2 |
| A�� | ��a������b��ϡ���ᣨ��������a������c��Һ�У���a��b��Ӧ�����ӷ���ʽΪFe+4H++NO3-=Fe3++NO��+2H2O | |
| B�� | ��c��dΪ���壬�Ҷ���ʹ����ʯ��ˮ����ǣ��˻������ͨ����ˮ�У���ɫ��ȥ������ɫ���̵����ӷ���ʽΪSO2+Br2+2H2O�T4H++SO${\;}_{4}^{2-}$+2Br- | |
| C�� | ��c����ɫ�̼�����ζ�����壬��ˮ��Һ�������ԣ��ڱ�״�������ſ������ռ�c�����ƽ��Ħ������Ϊ20 g��mol-'1�Ļ�����������Ȫʵ�飮�������ʲ���ɢ��ʵ����ɺ�������Һ�����ʵ���Ũ��ԼΪ0�� 056 mol��L-1 | |
| D�� | ��a���������ЧӦ����Ҫ����֮һ��c��d��Ϊ���Σ��μӷ�Ӧ��a��b���ʵ���֮��Ϊ4��5����������Ӧ�����ӷ���ʽΪ4CO2+5OH-�TCO${\;}_{3}^{2-}$+3HCO${\;}_{3}^{-}$+H2O |
 ѧУ��ѧ�о�С���ij��Һ���м�����������ɫ��Һ�п��ܺ���NH4+��K+��Al3+��HCO3-��Cl-��MnO4-��SO42-�������еļ������ӣ�
ѧУ��ѧ�о�С���ij��Һ���м�����������ɫ��Һ�п��ܺ���NH4+��K+��Al3+��HCO3-��Cl-��MnO4-��SO42-�������еļ������ӣ� ���ռ乹��Ϊ�����Σ�
���ռ乹��Ϊ�����Σ�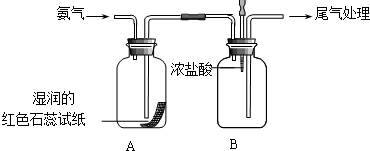 ijС��ͬѧ������ͼ��ʾװ��̽�����������ʣ�
ijС��ͬѧ������ͼ��ʾװ��̽�����������ʣ� ����ͼװ�ý���ʵ�飬��A��μ���B�У�
����ͼװ�ý���ʵ�飬��A��μ���B�У� ��ҵ��ⱥ��ʳ��ˮģ��װ�õĽṹ��ͼ��ʾ��
��ҵ��ⱥ��ʳ��ˮģ��װ�õĽṹ��ͼ��ʾ�� ��
��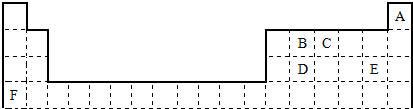
 ���Ƚ�E��F�����ּ����ӵİ뾶��С��E-���ڣ�����ڻ�С�ڣ�F+
���Ƚ�E��F�����ּ����ӵİ뾶��С��E-���ڣ�����ڻ�С�ڣ�F+