��Ŀ����
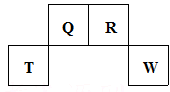
������Ԫ��Q��R��T��W��Ԫ�����ڱ��е�λ����ͼ��ʾ������T��������������������������ȣ���ش��������⣺

��1��T��ԭ�ӽṹʾ��ͼΪ ��
��2��Ԫ�صķǽ����ԣ�Q W���������������
��3��W�ĵ�����������������ˮ����Ũ��Һ�����ܷ�����Ӧ�������������ʣ�����һ�������壬��Ӧ�Ļ�ѧ����ʽΪ ��
��4��R�ж�����������м���Է���������С����һ�������£�2L�ļ�������0.5L���������ϣ����û�����屻������NaOH��Һ��ȫ���պ�û����������������ɵ�R�ĺ������εĻ�ѧʽ�� ��


��1��T��ԭ�ӽṹʾ��ͼΪ ��
��2��Ԫ�صķǽ����ԣ�Q W���������������
��3��W�ĵ�����������������ˮ����Ũ��Һ�����ܷ�����Ӧ�������������ʣ�����һ�������壬��Ӧ�Ļ�ѧ����ʽΪ ��
��4��R�ж�����������м���Է���������С����һ�������£�2L�ļ�������0.5L���������ϣ����û�����屻������NaOH��Һ��ȫ���պ�û����������������ɵ�R�ĺ������εĻ�ѧʽ�� ��
��1�� ����2������ ��3��S��2H2SO4(Ũ)
����2������ ��3��S��2H2SO4(Ũ)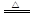 3SO2����2H2O ��4��NaNO2
3SO2����2H2O ��4��NaNO2
 ����2������ ��3��S��2H2SO4(Ũ)
����2������ ��3��S��2H2SO4(Ũ)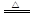 3SO2����2H2O ��4��NaNO2
3SO2����2H2O ��4��NaNO2��������Ԫ�������ڱ��е����λ�ÿ��Կ�����Tһ�����ڵ������ڡ�����ΪT��������������������������ȣ�����Tֻ����AlԪ�أ����Q��R��W����ΪC��N��SԪ�ء���N����������NO����Է���������С���μӷ�Ӧ��NO��O�����ʵ���֮��4�U1�����ݵ�ʧ�����غ��֪1molNO�ڷ�ӦӦʧȥ1mol���ӣ������ϼ��ɣ�2�����ߵ���3�ۣ����Ժ������εĻ�ѧʽ��NaNO2

��ϰ��ϵ�д�
�����Ŀ