��Ŀ����
��18�֣���֪��
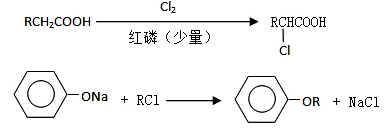
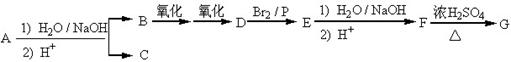
I������ƽF�ǽ�Ѫ֬�������̴���ҩ�����һ���ϳ�·�����£�
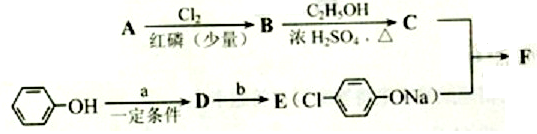
��1��AΪ����һԪ���ᣬ8.8g A������NaHCO3��Һ��Ӧ����2.24L CO2����״������A�ķ���ʽΪ___________________��
��2��д������A����ʽ�����м������Ľṹ��ʽ�� ______________________________��
��3��B���ȴ����ᣬ��˴Ź��������������壬д��B��C�ķ�Ӧ����ʽ��
__________________________________________________________��
��4��C+E��F�ķ�Ӧ����Ϊ________________________��
��5��д��A��F�Ľṹ��ʽ��A______________________�� F__________________________��
��6��D�ı������������⣬�����������ŵ�����Ϊ___________________��д��a��b���������Լ���a ______________; b___________��
��. ������·�ߣ���C�ɺϳɸ߾���H��
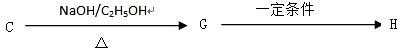
��7��C G�ķ�Ӧ����Ϊ_____________________.
G�ķ�Ӧ����Ϊ_____________________.
��8��д��G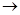 H�ķ�Ӧ����ʽ��_______________________��
H�ķ�Ӧ����ʽ��_______________________��

I������ƽF�ǽ�Ѫ֬�������̴���ҩ�����һ���ϳ�·�����£�

��1��AΪ����һԪ���ᣬ8.8g A������NaHCO3��Һ��Ӧ����2.24L CO2����״������A�ķ���ʽΪ___________________��
��2��д������A����ʽ�����м������Ľṹ��ʽ�� ______________________________��
��3��B���ȴ����ᣬ��˴Ź��������������壬д��B��C�ķ�Ӧ����ʽ��
__________________________________________________________��
��4��C+E��F�ķ�Ӧ����Ϊ________________________��
��5��д��A��F�Ľṹ��ʽ��A______________________�� F__________________________��
��6��D�ı������������⣬�����������ŵ�����Ϊ___________________��д��a��b���������Լ���a ______________; b___________��
��. ������·�ߣ���C�ɺϳɸ߾���H��
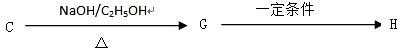
��7��C
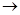 G�ķ�Ӧ����Ϊ_____________________.
G�ķ�Ӧ����Ϊ_____________________.��8��д��G
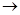 H�ķ�Ӧ����ʽ��_______________________��
H�ķ�Ӧ����ʽ��_______________________����1��C4H8O2�� ��2��HCOOCH2CH2CH3 HCOOCH(CH3)2��
��3��(CH3)2CClCOOH+C2H5OH (CH3)2CClCOOC2H5+H2O��
(CH3)2CClCOOC2H5+H2O��
��4��ȡ����Ӧ ��5��(CH3)2CHCOOH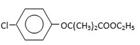
��6��Cl2 NaOH��Һ ��7����ȥ��Ӧ
��8��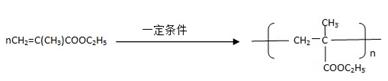
��3��(CH3)2CClCOOH+C2H5OH
 (CH3)2CClCOOC2H5+H2O��
(CH3)2CClCOOC2H5+H2O����4��ȡ����Ӧ ��5��(CH3)2CHCOOH

��6��Cl2 NaOH��Һ ��7����ȥ��Ӧ
��8��

��1���ɷ���ʽRCOOH+NaHCO3=RCOONa+H2O+CO2����֪����2.24L CO2����״��������Ҫ����0.1mol���ᣬ��A��Ħ��������88g/mol����Ϊ�Ȼ���COOH��Ħ��������45g/mol����������������Ħ��������43g/mol����������Ļ�ѧʽ��C3H7����A�ķ���ʽ��C4H8O2��
��2������ʽ��C4H8O2�ļ�����˵�����ɼ���ͱ�����ʽ�ģ�����������������������֣���������Ӧ�ļ�����Ҳ�����֣��ֱ�Ϊ��HCOOCH2CH2CH3��HCOOCH(CH3)2��
��3�������Ȼ��к���һ����ԭ�ӣ�˵���������ԭ��ȫ�����ڼ����Ҽ�����ͬһ��̼ԭ���ϣ���B�Ľṹ��ʽ��(CH3)2CClCOOH�����A�Ľṹ��ʽ��(CH3)2CHCOOH������B��C�ķ�Ӧ����ʽΪ:
(CH3)2CClCOOH+C2H5OH (CH3)2CClCOOC2H5+H2O��
(CH3)2CClCOOC2H5+H2O��
��4������Ϣ��֪C��E��Ӧ�ķ���ʽΪ��
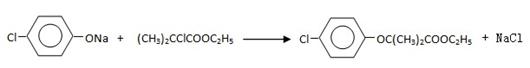 ����˸÷�Ӧ����ȡ����Ӧ��
����˸÷�Ӧ����ȡ����Ӧ��
��5����������3���ͣ�4����
��6�����ݺϳ�·�߿�֪D�Ľṹ��ʽ��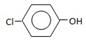 ������a��b���������Լ��ֱ���������NaOH��Һ��
������a��b���������Լ��ֱ���������NaOH��Һ��
��7��C�Ľṹ��ʽΪ(CH3)2CClCOOC2H5��������ԭ�ӣ��������������ƵĴ���Һ�п��Է�����ȥ��Ӧ��������G�Ľṹ��ʽ��CH2=C(CH3)COOC2H5��G�к���̼̼˫�������Է����Ӿ۷�Ӧ���ɸ߷��ӻ�����H������ʽΪ
 ��
��
��8����������7����
��2������ʽ��C4H8O2�ļ�����˵�����ɼ���ͱ�����ʽ�ģ�����������������������֣���������Ӧ�ļ�����Ҳ�����֣��ֱ�Ϊ��HCOOCH2CH2CH3��HCOOCH(CH3)2��
��3�������Ȼ��к���һ����ԭ�ӣ�˵���������ԭ��ȫ�����ڼ����Ҽ�����ͬһ��̼ԭ���ϣ���B�Ľṹ��ʽ��(CH3)2CClCOOH�����A�Ľṹ��ʽ��(CH3)2CHCOOH������B��C�ķ�Ӧ����ʽΪ:
(CH3)2CClCOOH+C2H5OH
 (CH3)2CClCOOC2H5+H2O��
(CH3)2CClCOOC2H5+H2O����4������Ϣ��֪C��E��Ӧ�ķ���ʽΪ��
 ����˸÷�Ӧ����ȡ����Ӧ��
����˸÷�Ӧ����ȡ����Ӧ����5����������3���ͣ�4����
��6�����ݺϳ�·�߿�֪D�Ľṹ��ʽ��
 ������a��b���������Լ��ֱ���������NaOH��Һ��
������a��b���������Լ��ֱ���������NaOH��Һ����7��C�Ľṹ��ʽΪ(CH3)2CClCOOC2H5��������ԭ�ӣ��������������ƵĴ���Һ�п��Է�����ȥ��Ӧ��������G�Ľṹ��ʽ��CH2=C(CH3)COOC2H5��G�к���̼̼˫�������Է����Ӿ۷�Ӧ���ɸ߷��ӻ�����H������ʽΪ
 ��
����8����������7����

��ϰ��ϵ�д�
�����Ŀ
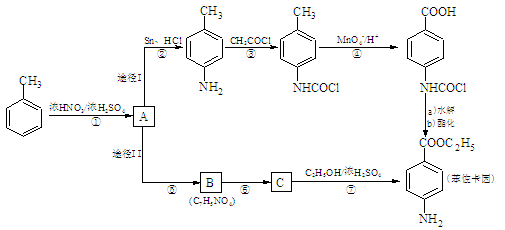
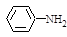 �������еİ����ױ�������
�������еİ����ױ������� ��
�� ��
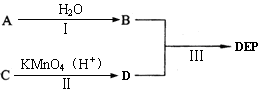
��
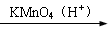
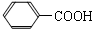
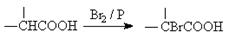
 2CH3COOH������AΪ��Ҫԭ�Ϻϳ�������������ϳ�·������ͼ��ʾ��
2CH3COOH������AΪ��Ҫԭ�Ϻϳ�������������ϳ�·������ͼ��ʾ��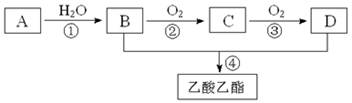
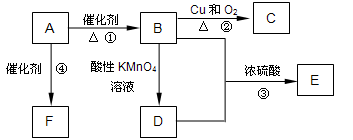
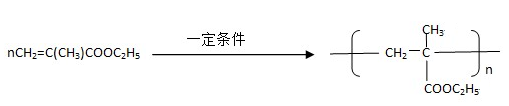
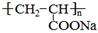 ����һ��ǿ��ˮ��֬����ij��A�ϳɾ۱�ϩ���Ƶ��������£�
����һ��ǿ��ˮ��֬����ij��A�ϳɾ۱�ϩ���Ƶ��������£�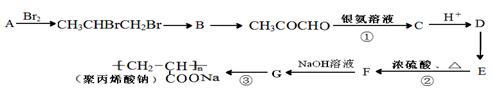
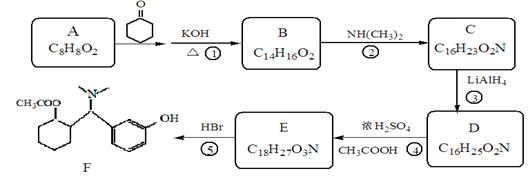
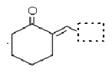
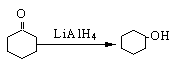
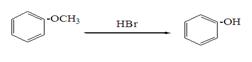
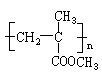 �ĵ������������ƹ��Ⱥ����ϵ�У�������������ܵõ������л�����еģ� �����������֮һ��Ϊͬ���칹�塣
�ĵ������������ƹ��Ⱥ����ϵ�У�������������ܵõ������л�����еģ� �����������֮һ��Ϊͬ���칹�塣