��Ŀ����
�ִ���п�ķ����ɷ�Ϊ��ʪ�������࣬������������п�ĸ���Ʒ�����ڸ߶��Խ������Իش�����������⣮
(1)����п�ǽ���п��(��Ҫ��ZnS)ͨ����ѡ������ʹ��ת��Ϊ����п���ٰ�����п�ͽ�̿��ϣ��ڹķ�¯�м�����1 373��1 573 K��ʹп�����������Ҫ��ӦΪ��2ZnS��3O2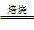 2ZnO��2SO2���ķ�¯�У�2C��O2
2ZnO��2SO2���ķ�¯�У�2C��O2 2CO���ķ�¯�У�ZnO��CO
2CO���ķ�¯�У�ZnO��CO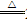
 Zn��CO2
Zn��CO2
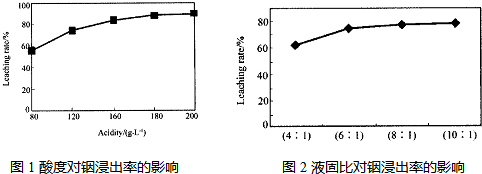
�ӻ���п�����IJ����к����ֽ������ʼ�In2O3������������ȡ����ij�о������Դ��о��������¡�ʵ�����漰�����(ÿ����Һ�к����������)�����Ľ���������ͼ1��������Һ��������������������ͼ2��
(1)����п�ǽ���п��(��Ҫ��ZnS)ͨ����ѡ������ʹ��ת��Ϊ����п���ٰ�����п�ͽ�̿��ϣ��ڹķ�¯�м�����1 373��1 573 K��ʹп�����������Ҫ��ӦΪ��2ZnS��3O2
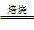 2ZnO��2SO2���ķ�¯�У�2C��O2
2ZnO��2SO2���ķ�¯�У�2C��O2 2CO���ķ�¯�У�ZnO��CO
2CO���ķ�¯�У�ZnO��CO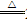
 Zn��CO2
Zn��CO2�ӻ���п�����IJ����к����ֽ������ʼ�In2O3������������ȡ����ij�о������Դ��о��������¡�ʵ�����漰�����(ÿ����Һ�к����������)�����Ľ���������ͼ1��������Һ��������������������ͼ2��
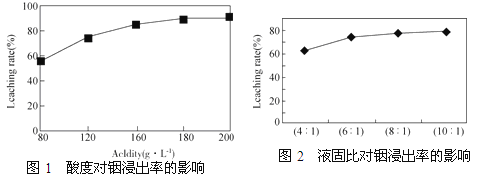
�ٵ����Ϊ196ʱ�������ʵ���Ũ��Ϊ________��
�ڴӽ�Լԭ�Ϻͽ����ʿ��ǣ����˵���Ⱥ�Һ�̱ȷֱ�Ϊ��____________��__________��
(2)ʪ����п����Ҫ��������Ϊ

�������ȡ����Ҫ��Ӧ�����ӷ���ʽΪ__________��
�ڴӱ��������ͳ������ԭ�ϽǶȣ���δ���������������__________��
�۳�ȥ�����Һ�е���������H2O2�������ٵ���pHʹ֮�γ�Fe(OH)3������д��H2O2����Fe2�������ӷ���ʽ��__________��
�������Һ������Cd2����Ϊ�˷�ֹ����Ⱦ�������ӣ������������ʵIJ��죬��������������Һ���룬��֪Zn(OH)2����������һ��Ҳ�������ԣ���д����������ӷ���ʽ��________________��________________��
�ڴӽ�Լԭ�Ϻͽ����ʿ��ǣ����˵���Ⱥ�Һ�̱ȷֱ�Ϊ��____________��__________��
(2)ʪ����п����Ҫ��������Ϊ

�������ȡ����Ҫ��Ӧ�����ӷ���ʽΪ__________��
�ڴӱ��������ͳ������ԭ�ϽǶȣ���δ���������������__________��
�۳�ȥ�����Һ�е���������H2O2�������ٵ���pHʹ֮�γ�Fe(OH)3������д��H2O2����Fe2�������ӷ���ʽ��__________��
�������Һ������Cd2����Ϊ�˷�ֹ����Ⱦ�������ӣ������������ʵIJ��죬��������������Һ���룬��֪Zn(OH)2����������һ��Ҳ�������ԣ���д����������ӷ���ʽ��________________��________________��
(1)��2 mol/L��
��180����6��1
(2)��ZnO��2H��===Zn2����H2O
�ڽ��̵����������������������
��2Fe2����H2O2��2H��===2Fe3����2H2O
��Cd2����2OH��===Cd(OH)2�� ��Zn2����4OH��===ZnO22����2H2O[��Zn2����4OH��===Zn(OH)42��]
��180����6��1
(2)��ZnO��2H��===Zn2����H2O
�ڽ��̵����������������������
��2Fe2����H2O2��2H��===2Fe3����2H2O
��Cd2����2OH��===Cd(OH)2�� ��Zn2����4OH��===ZnO22����2H2O[��Zn2����4OH��===Zn(OH)42��]

��ϰ��ϵ�д�
 �̲�ȫ���ִʾ�ƪϵ�д�
�̲�ȫ���ִʾ�ƪϵ�д�
�����Ŀ


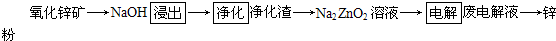



 2ZnO+2SO2
���ķ�¯�У�2C +O2
2ZnO+2SO2
���ķ�¯�У�2C +O2 2CO
2CO

