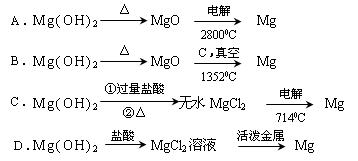
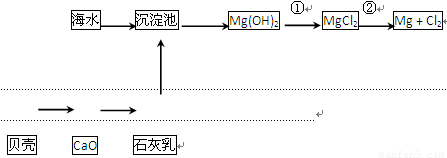
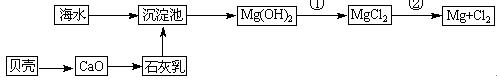
��Ŀ����
��10�֣�
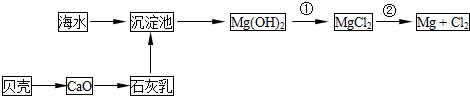
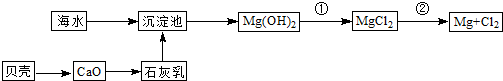
�Ӻ�ˮ�п��Ի�õ�ˮ��ʳ�β�����ȡþ��������ʡ�
��1����ˮ�����ķ�����Ҫ�� ����һ�֣���
��2��������ѧ�仯���ܴӺ�ˮ�л�õ������� ������ţ�
| A���ȡ��塢�� | B���ơ�þ���� | C���ռ���� | D��ʳ�Ρ���ˮ |
��Ӧ�ٵ����ӷ���ʽΪ ��
��Ӧ�ڵĻ�ѧ����ʽΪ ��
��4���Ӻ�ˮ����ȡ�����Ҫ��������Ũ���ĺ�ˮ��ͨ�����������������������÷�Ӧ�����ӷ���ʽΪ ��
��10�֣�
��1�������������������ӽ�������2�֣�
��2��ABC��2�֣�
��3��Mg(OH)2+2H+ Mg2++H2O��2�֣���MgCl2(����)
Mg2++H2O��2�֣���MgCl2(����)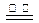 Mg+Cl2����2�֣�
Mg+Cl2����2�֣�
��4��Cl2+2Br�� 2Cl��+Br2��2�֣�
2Cl��+Br2��2�֣�
����

��ϰ��ϵ�д�
�����Ŀ
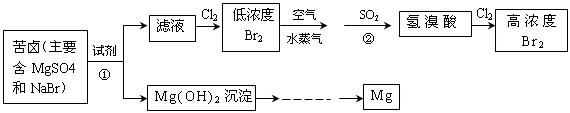
 ʱ��һ��ͨ��Cl
ʱ��һ��ͨ��Cl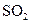 ���� �����������ԭ�����ԣ�
���� �����������ԭ�����ԣ�