��Ŀ����
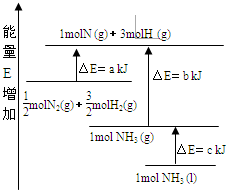
��ѧ��ӦN2+3H2��2NH3�������仯��ͼ��ʾ��E����ֵ���÷�Ӧ���Ȼ�ѧ����ʽ��( �� )
| A��N2(g)+3H2(g)��2NH3(1)�� ��H��2(a-b-c)kJ��mol��1 |
| B��N2(g)+3H2(g)��2NH3(g)����H��2(b-a)kJ��mol��1 |
C�� N2(g)+ N2(g)+ H2(g)��NH3(1)����H��(b+c-a)kJ��mol��1 H2(g)��NH3(1)����H��(b+c-a)kJ��mol��1 |
D�� N2(g)+ N2(g)+ H2(g)��NH3(g)�� ��H��(a+b)kJ��mol H2(g)��NH3(g)�� ��H��(a+b)kJ��mol |
A
�����������������ͼ���֪����Ӧ��������������������������������������Ӧ�Ƿ��ȷ�Ӧ��A��ȷ��C����ȷ������������ǰ�������Ӧ���Ȼ�ѧ����ʽ��N2(g)+3H2(g)��2NH3(g)����H����2(b-a)kJ��mol��1�����B��D���Ǵ���ģ���ѡA��
���㣺���鷴Ӧ�ȵ��жϡ����㼰ͼ��ʶ��
�����������Ǹ߿��еij���ͼ��������ͼ�������������������Ѷȡ���������������ѧ����ͼ��ʶ�����������Ӧ������������������ѧ��Ӧ�����������ѧ����ѧϰЧ�ʡ�

��ϰ��ϵ�д�
 ����ѵ��ϵ�д�
����ѵ��ϵ�д� ��ĩ�����ϵ�д�
��ĩ�����ϵ�д�
�����Ŀ
 ��ѧ��ӦN2+3H2��2NH3�������仯��ͼ��ʾ���÷�Ӧ���Ȼ�ѧ����ʽ�ǣ�������
��ѧ��ӦN2+3H2��2NH3�������仯��ͼ��ʾ���÷�Ӧ���Ȼ�ѧ����ʽ�ǣ�������| A��N2��g��+H2��g����NH3��1��-46 kJ | B��N2��g��+H2��g����NH3��g��-454 kJ | C��N2��g��+3 H2��g����2 NH3��g��+92 kJ | D��N2��g��+3 H2��g����2 NH3��1��+431.3 kJ |
 ��ѧ��ӦN2+3H2?2NH3�������仯��ͼ��ʾ��E����ֵ���÷�Ӧ���Ȼ�ѧ����ʽ�ǣ�������
��ѧ��ӦN2+3H2?2NH3�������仯��ͼ��ʾ��E����ֵ���÷�Ӧ���Ȼ�ѧ����ʽ�ǣ�������| A��N2��g��+3H2��g��?2NH3��l������H=2��a-b-c��kJ?mol-1 | ||||
| B��N2��g��+3H2��g��?2NH3��g������H=2��b-a��kJ?mol-1 | ||||
C��
| ||||
D��
|
 ��2008?���죩��ѧ��ӦN2+3H2=2NH3�������仯��ͼ��ʾ���÷�Ӧ���Ȼ�ѧ����ʽ�ǣ�������
��2008?���죩��ѧ��ӦN2+3H2=2NH3�������仯��ͼ��ʾ���÷�Ӧ���Ȼ�ѧ����ʽ�ǣ�������