��Ŀ����
�ϳ�����CO��H2����һ����Ҫ�Ļ���ԭ�ϣ��ڻ��������о���ʮ�ֹ㷺����;�����Ʊ������õ�һ�ֹ���������ͼ��ʾ������X��X�����Ҫ�ɷ����ƣ���
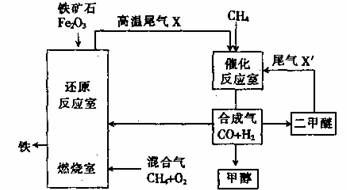
��1���ںϳ���ұ���������������У���ȼ������ͨ�������������Ŀ���� �������������V��CH4����V��O2��Ϊ ��
��2���״��������ѣ�CH3OCH3�������ʱ���Ϊ21���͵�����ȼ�ϣ�������ࡢ��Ч�����������ܡ�
��д���úϳ��������״��Ļ�ѧ����ʽ�� ���ø÷����ϳɼ״�������ŵ��� ��
���úϳ���Ϊԭ�����������ѣ�����ԭ����ȵIJ�ͬ��Ӧ���ﲻ����ͬ����������β����ѭ��ʹ�ã���д����CO��H2Ϊԭ����ȡ�����ѵ����ֲ�ͬ�Ļ�ѧ����ʽ�� ��
��
��3��ij��������ͼʹ����ͼ��ʾ�������������״��������ѡ�����������Ϊ�Ƿ���У��������У���˵�����ɣ������У�����������ʲô�ŵ㣿
��
��1��Ϊ��ԭ����ʯ�ṩ���� 1��2
��2����CO��2H2 CH3OH�� ԭ��������Ϊ100%
��2CO��4 H2 CH3OCH3��H2O��3CO��3H2 CH3OCH3��CO2
��3������ ��Ϊԭ���á�ԭ�������ʸߡ��м��������Ч��ѭ�����á���Ʒ�ṹ�Ķ�����

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�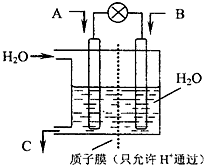 ��2010?��������ģ�����ȫ����ڵ������쳣�����������ٴγ�Ϊ���㣮SO2��NOx��CO2�ǶԻ���Ӱ��ϴ�����壬�����ǵĺ������ƺ��������Ż��������滷������Ч;����
��2010?��������ģ�����ȫ����ڵ������쳣�����������ٴγ�Ϊ���㣮SO2��NOx��CO2�ǶԻ���Ӱ��ϴ�����壬�����ǵĺ������ƺ��������Ż��������滷������Ч;���� �ϳ�����CO��H2������Ҫ�Ļ���ԭ�ϣ��ڹ�ҵ���������Ź㷺��Ӧ�ã�
�ϳ�����CO��H2������Ҫ�Ļ���ԭ�ϣ��ڹ�ҵ���������Ź㷺��Ӧ�ã�