��Ŀ����
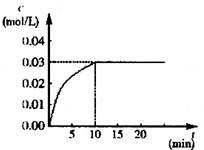
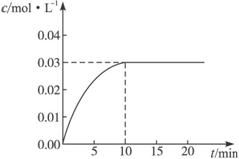
��ijһ�ݻ�Ϊ5 L���ܱ������ڣ�����0��2 mol��CO��0��2 mol��H2O���ڴ������ڵ������¸��¼��ȣ��������·�Ӧ��CO(g)��H2O(g) CO2(g)��H2(g)������ӦΪ���ȷ�Ӧ����Ӧ��CO2��Ũ����ʱ��仯�����ͼ
CO2(g)��H2(g)������ӦΪ���ȷ�Ӧ����Ӧ��CO2��Ũ����ʱ��仯�����ͼ
 CO2(g)��H2(g)������ӦΪ���ȷ�Ӧ����Ӧ��CO2��Ũ����ʱ��仯�����ͼ
CO2(g)��H2(g)������ӦΪ���ȷ�Ӧ����Ӧ��CO2��Ũ����ʱ��仯�����ͼ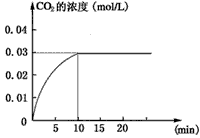
��1������ͼ�����ݣ���Ӧ��ʼ���ﵽƽ��ʱ��CO��ƽ����Ӧ����Ϊ__________����Ӧ�ﵽƽ��ʱ��
c(H2)��__________________��
��2���жϸ÷�Ӧ�ﵽƽ���������__________��
��CO���ٵĻ�ѧ��Ӧ���ʺ�CO2���ٵĻ�ѧ��Ӧ�������
��CO��H2O��CO2��H2��Ũ�ȶ����
��CO��H2O��CO2��H2��Ũ�ȶ����ٷ����仯
�������淴Ӧ���ʶ�Ϊ��
c(H2)��__________________��
��2���жϸ÷�Ӧ�ﵽƽ���������__________��
��CO���ٵĻ�ѧ��Ӧ���ʺ�CO2���ٵĻ�ѧ��Ӧ�������
��CO��H2O��CO2��H2��Ũ�ȶ����
��CO��H2O��CO2��H2��Ũ�ȶ����ٷ����仯
�������淴Ӧ���ʶ�Ϊ��
��1��0��003mol/(L��min)��0��03mol/L
��2���٢�
��2���٢�

��ϰ��ϵ�д�
�����Ŀ
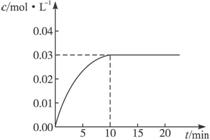
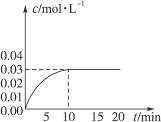
��ijһ�ݻ�Ϊ5 L���ܱ������ڣ�����0.3 mol��CO��0.3 mol��H2O���ڴ������ں�800��������¼��ȣ��������·�Ӧ��CO(g)��H2O(g)  CO2(g)��H2(g) ��H>0����Ӧ��CO2��Ũ����ʱ��仯�������ͼ��
CO2(g)��H2(g) ��H>0����Ӧ��CO2��Ũ����ʱ��仯�������ͼ��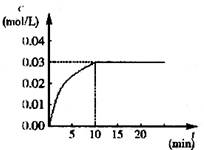
(1)����ͼ�����ݣ���Ӧ��ʼ���ﵽƽ��ʱ��
CO�Ļ�ѧ��Ӧ����Ϊv(CO)�� mol/(L��min)-1��
���¶��µ�ƽ�ⳣ��K�� ��
(2)���������������£��ı������������´ﵽƽ��ʱ��ʹƽ�ⳣ��K�������
������ĸ��
| A�������¶� | B�������¶� | C������ѹǿ | D����Сѹǿ |
(3)��Ҫһ��ʼ����0.1 mol��CO��0.1 mol��H2O��0.2 mol��CO2��0.2 mol��H2������ͬ�������£���Ӧ��ƽ��ʱ��c(CO)��________________mol/L.
(4)�������¶Ⱥ�������������䣬��(1)������ƽ����ϵ�У��ٳ���0.3mol ��ˮ���������´ﵽƽ���CO��ת����_______(����ߡ��������͡����ǡ����䡱)��CO2����������________(����ߡ��������͡����ǡ����䡱).
(5)�ڴ������ں�800��������£���ijһʱ�̲��C(CO)=C(H2O)= 0.09mol/L,
C(CO2 )=C(H2)=0.13mol/L ,��˷�Ӧ�Ƿ���ƽ��״̬: (��ǡ���),��û�д���ƽ��״̬��÷�Ӧ�� �ƶ�������������淽��
 CO2(g)��H2(g)
��H>0����Ӧ��CO2��Ũ����ʱ��仯�������ͼ��
CO2(g)��H2(g)
��H>0����Ӧ��CO2��Ũ����ʱ��仯�������ͼ��