��Ŀ����
�����£���CaCl2��������ɵĻ����Һ500 mL�У���֪c(Ca2+)=0.3 mol/L��c(Cl -)=1.6 mol��L����Һ�ܶ�ԼΪ1.A.�٢ۢ� B.�ڢ� C.�ڢۢ� D.�٢�
B
�����������ۺϿ�������Һ���֪ʶ������ҺpH����Һ���ʵ���Ũ�ȡ��������������ȡ��ɵ���غ�n(Ca2+)��2+n(H+)=n(Cl-)+n(OH-)��ˮ�����ӻ�c(H+)��c(OH-)=10-14�ɵã�c(H+)=1 mol��L��c(OH-)=10-14 mol��L��n(Ca2+)=0.3 mol��L��0.![]()
=![]() ��100��=1.1����
��100��=1.1����

��ϰ��ϵ�д�
 ��ĩ�����ϵ�д�
��ĩ�����ϵ�д�
�����Ŀ
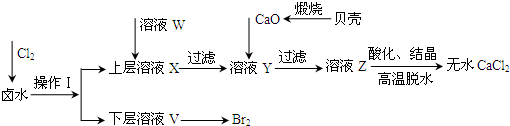
 �ס��ҡ������������ɶ�����Ԫ����ɵĵ��ʣ�X��Y��Z��W��Ϊ����������£�XΪ����ɫ���壬Z��������Ϊ����ɫ��W��ʹʪ��ĺ�ɫʯ����ֽ����ɫ����ҵ�ϳ���W����ȡZ����ת����ϵ����ͼ��ʾ��
�ס��ҡ������������ɶ�����Ԫ����ɵĵ��ʣ�X��Y��Z��W��Ϊ����������£�XΪ����ɫ���壬Z��������Ϊ����ɫ��W��ʹʪ��ĺ�ɫʯ����ֽ����ɫ����ҵ�ϳ���W����ȡZ����ת����ϵ����ͼ��ʾ��