��Ŀ����
��11�֣�ijѧ����0.2000mol��L��1�ı�NaOH��Һ�ζ�δ֪Ũ�ȵ����ᣬ������ɷ�Ϊ���¼�����
��������ˮϴ�Ӽ�ʽ�ζ��ܣ�ע��0.2000mol��L��1�ı�NaOH��Һ����0���̶�������
�ڹ̶��õζ��ܲ�ʹ�ζ��ܼ������Һ�壻
�۵���Һ������0����0���̶������£������¶�����
����ȡ20.00mL����Һע��ྻ����ƿ�У�������3�η�̪��Һ��
���ñ�Һ�ζ����յ㣬���µζ���Һ�������
���ظ����ϵζ�����2-3�Ρ�
��ش�
(1)���ϲ����д�����ǣ����ţ�________���ô�������ᵼ�²ⶨ���_________
(�ƫ����ƫС������Ӱ�족)��
(2)������У���ȡ20.00mL����ҺӦʹ��__________________(����������)������ƿװҺǰ��������������ˮ���ⶨ���____________�������ƫС������Ӱ�족����
(3)����ݵζ�ʱ�۾�Ӧע�� _______________________________�жϵ���ζ��յ�������ǣ�__________________________________________________��
(4)������ʵ�����ݼ�¼��
���ϱ����Կ�������1�εζ���¼��NaOH��Һ������Զ��ں����ε����������ܵ�ԭ���ǣ� ��
A���ζ�ǰ�ζ��ܼ��������ݣ��ζ�����������
B����ƿ�ô���Һ��ϴ
C��NaOH��Һ����ʱ��������в��ֱ���
D���ζ�����ʱ�����Ӽ���
(5) �����ϱ���¼���ݣ�ͨ������ɵã�������Ũ��Ϊ��____________mol��L��1
��������ˮϴ�Ӽ�ʽ�ζ��ܣ�ע��0.2000mol��L��1�ı�NaOH��Һ����0���̶�������
�ڹ̶��õζ��ܲ�ʹ�ζ��ܼ������Һ�壻
�۵���Һ������0����0���̶������£������¶�����
����ȡ20.00mL����Һע��ྻ����ƿ�У�������3�η�̪��Һ��
���ñ�Һ�ζ����յ㣬���µζ���Һ�������
���ظ����ϵζ�����2-3�Ρ�
��ش�
(1)���ϲ����д�����ǣ����ţ�________���ô�������ᵼ�²ⶨ���_________
(�ƫ����ƫС������Ӱ�족)��
(2)������У���ȡ20.00mL����ҺӦʹ��__________________(����������)������ƿװҺǰ��������������ˮ���ⶨ���____________�������ƫС������Ӱ�족����
(3)����ݵζ�ʱ�۾�Ӧע�� _______________________________�жϵ���ζ��յ�������ǣ�__________________________________________________��
(4)������ʵ�����ݼ�¼��
| �ζ����� | ���������mL�� | NaOH��Һ���������mL�� | |
| �ζ�ǰ | �ζ��� | ||
| 1 | 20.00 | 0.00 | 18.10 |
| 2 | 20.00 | 0.00 | 16.30 |
| 3 | 20.00 | 0.00 | 16.22 |
A���ζ�ǰ�ζ��ܼ��������ݣ��ζ�����������
B����ƿ�ô���Һ��ϴ
C��NaOH��Һ����ʱ��������в��ֱ���
D���ζ�����ʱ�����Ӽ���
(5) �����ϱ���¼���ݣ�ͨ������ɵã�������Ũ��Ϊ��____________mol��L��1
��11�֣���1���٣�1�֣�ƫ��1�֣�
��2����ʽ�ζ��ܣ�����Һ�ܣ���1�֣� ��Ӱ�죨1�֣�
��3����ƿ����Һ��ɫ�仯��1�֣�
��ƿ����Һ����ɫ��Ϊdz��ɫ��1�֣�������Ӳ���ɫ��1�֣�
��4��A B��2�֣���5��0.1626��2�֣�
��2����ʽ�ζ��ܣ�����Һ�ܣ���1�֣� ��Ӱ�죨1�֣�
��3����ƿ����Һ��ɫ�仯��1�֣�
��ƿ����Һ����ɫ��Ϊdz��ɫ��1�֣�������Ӳ���ɫ��1�֣�
��4��A B��2�֣���5��0.1626��2�֣�
��1��ʢװ��Һ�ļ�ʽ�ζ��ܱ�����NaOH��Һ��ϴ�������±�Һ��Ũ��ƫС�������ı�Һ�����ƫ���²ⶨ���ƫ��
��2����ȷ��ȡ����Һ���Ӧѡ�ýϾ�����������ʽ�ζ��ܣ�����Һ�ܣ�
��ƿ�м�ʹ������ˮ�������ܸı���Һ��H+���������ʶԲⶨ�����Ӱ��
��3���ζ�ʱ�۾�Ӧ�۲���ƿ����Һ��ɫ�仯��
����Һ�����Ե��ζ��յ�ʱ��ָʾ����̪��ɫ����ɫת��Ϊ�ۺ�ɫ���Ұ���Ӳ���ɫ
��4��A����Һռ�������ݵ����������û�����ڵζ�������Һ���ƫ��
B����ƿ�ô���Һ��ϴ�������������ṩ��H+ƫ�࣬�����ı�Һ���ƫ��
C���ζ��յ�ʱ����ҺΪNaCl��Һ����ʹNaOH�����տ����е�CO2�����ֱ��ʣ�������Ӱ�����ñ�Һ���
D���ζ�����ʱ�����Ӽ��������±�Һ�����ƫС
(5)���ڵ�1�εζ���¼��NaOH��Һ������Զ��ں����ε��������ֻ�������α�Һ�����ƽ��ֵ����16.26ml��c(HCl)��0.02=0.2��0.01626����c(HCl)=0.1626mol��L��1
��2����ȷ��ȡ����Һ���Ӧѡ�ýϾ�����������ʽ�ζ��ܣ�����Һ�ܣ�
��ƿ�м�ʹ������ˮ�������ܸı���Һ��H+���������ʶԲⶨ�����Ӱ��
��3���ζ�ʱ�۾�Ӧ�۲���ƿ����Һ��ɫ�仯��
����Һ�����Ե��ζ��յ�ʱ��ָʾ����̪��ɫ����ɫת��Ϊ�ۺ�ɫ���Ұ���Ӳ���ɫ
��4��A����Һռ�������ݵ����������û�����ڵζ�������Һ���ƫ��
B����ƿ�ô���Һ��ϴ�������������ṩ��H+ƫ�࣬�����ı�Һ���ƫ��
C���ζ��յ�ʱ����ҺΪNaCl��Һ����ʹNaOH�����տ����е�CO2�����ֱ��ʣ�������Ӱ�����ñ�Һ���
D���ζ�����ʱ�����Ӽ��������±�Һ�����ƫС
(5)���ڵ�1�εζ���¼��NaOH��Һ������Զ��ں����ε��������ֻ�������α�Һ�����ƽ��ֵ����16.26ml��c(HCl)��0.02=0.2��0.01626����c(HCl)=0.1626mol��L��1

��ϰ��ϵ�д�
�����Ŀ
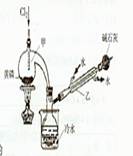
 5.00mL���������c1mol��L-lV1mL����Һ����ַ�Ӧ������c2mol��L-1Na2S2O3��Һ�ζ������ĵ⣬�յ�ʱ����V2mLNa2S2O3��Һ��
5.00mL���������c1mol��L-lV1mL����Һ����ַ�Ӧ������c2mol��L-1Na2S2O3��Һ�ζ������ĵ⣬�յ�ʱ����V2mLNa2S2O3��Һ��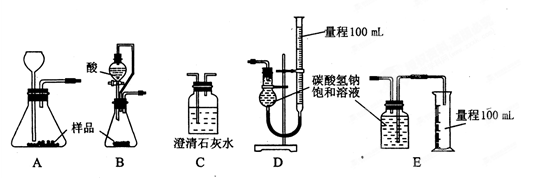
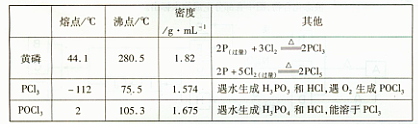
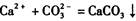 �ķ�Ӧ��������·���:��ȡ��Ʒ�������Һ����������ŨCaCl2��Һ����ַ�Ӧ����ˡ�ϴ�ӡ���������������㴿�ȡ�
�ķ�Ӧ��������·���:��ȡ��Ʒ�������Һ����������ŨCaCl2��Һ����ַ�Ӧ����ˡ�ϴ�ӡ���������������㴿�ȡ�
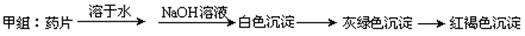 ����ͬѧ������Ƶķ������ʵ�飬���ź���������û�еõ�Ԥ�ڵ�ʵ������
����ͬѧ������Ƶķ������ʵ�飬���ź���������û�еõ�Ԥ�ڵ�ʵ������ 
 ����ʵ����֤��________________________________
����ʵ����֤��________________________________