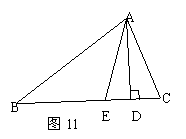
��Ŀ����
��֪����ͼ�٣��� �У�
�� ��
�� ��
�� ����
���� ��
�� ������
������ �������
������� �����˶����ٶ�Ϊ1cm/s����
�����˶����ٶ�Ϊ1cm/s���� ��
�� ������
������ �������
������� ��
��
���˶����ٶ�Ϊ2cm/s������ �������˶���ʱ��Ϊ
�������˶���ʱ��Ϊ ��
�� ��������������⣺
��������������⣺
��1���� Ϊ��ֵʱ��
Ϊ��ֵʱ�� ��
��
��2���� �����Ϊ
�����Ϊ ��
�� ������
������ ��
�� ֮��ĺ�����ϵʽ��
֮��ĺ�����ϵʽ��
��3���Ƿ����ijһʱ�� ��ʹ�߶�
��ʹ�߶� ǡ�ð�
ǡ�ð� ���ܳ������ͬʱƽ�֣������ڣ������ʱ
���ܳ������ͬʱƽ�֣������ڣ������ʱ ��ֵ���������ڣ�˵�����ɣ�
��ֵ���������ڣ�˵�����ɣ�
��4����ͼ�ڣ����� ������
������ ��
�� ���ۣ��õ��ı���
���ۣ��õ��ı��� ����ô�Ƿ����ijһʱ��
����ô�Ƿ����ijһʱ�� ��ʹ�ı���
��ʹ�ı��� Ϊ���Σ������ڣ������ʱ���εı߳����������ڣ�˵�����ɣ�
Ϊ���Σ������ڣ������ʱ���εı߳����������ڣ�˵�����ɣ�

��1��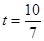 ����2��
����2��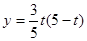 ����3�������ڣ���4��
����3�������ڣ���4��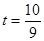 ��
��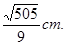
��������
�����������1����PQ��BCʱ���ɵó�������APQ��������ABC���ƣ��������������εĶ�Ӧ�߳ɱ���������ý����
��2����������APQ�������Ҫ��ȷ���ױߺߵ�ֵ���ױ�AQ���Ը���Q���ٶȺ�ʱ��t��ʾ�������ؼ��Ǹߣ�������AP�͡�A������ֵ����AP�ij�������AB-BP��ã���sinA����BC��AB��ֵ����˱�ʾ��AQ��AQ���ϵĸߺͿ��Եó�x��y�ĺ�����ϵʽ��
��3�������������ABC���ܳ������ƽ�֣���ôAP+AQ=BP+BC+CQ����ô������t��ʾ��CQ��AQ��AP��BP�ij�����ô���������ʱt��ֵ�����ǿɽ�t��ֵ���루2���������t�Ĺ�ϵʽ�У������ʱ����Ƕ��٣�Ȼ������Ƿ���������ABC�����һ�룬�Ӷ��жϳ��Ƿ������һʱ�̣�
��4�����ǿ�ͨ��������������������⣮����P��PM��AC��M��PN��BC��N����ôPNCM���Ǹ����Σ�����˼·��ͨ��������BPN��������ABC���ƣ��ó�����BP��PN��AB��AC�ı�����ϵ��������t��ʾ��PN�ij���Ҳ�ͱ�ʾ����MC�ij���Ҫ��ʹ�ı���PQP'C�����Σ�PQ=PC�����ݵ������������ߺ�һ���ص㣬QM=MC����������t��ʾ����AQ��QM��MC�����߶κ�AC�ij����Ϳ��Ը���AC=AQ+QM+MC�����t��ֵ�������t�Ϳ��Եó�QM��CM��PM�ij���Ҳ����������εı߳��ˣ�
��1����Rt��ABC�� ��
��
������֪��AP=5-t��AQ=2t����PQ��BC�����APQ�ס�ABC��
 ��
��
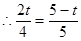
���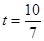 ��
��
���Ե�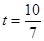 ʱ��PQ��BC��
ʱ��PQ��BC��
��2����ͼ������P��PH��AC��H��

�ߡ�APH�ס�ABC��
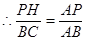 ��
��
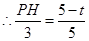
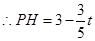 ��
��
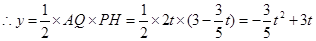 ��
��
��3����PQ�ѡ�ABC�ܳ�ƽ�֣���AP+AQ=BP+BC+CQ��
�ࣨ5-t��+2t=t+3+��4-2t�������t=1��
��PQ�ѡ�ABC���ƽ�֣���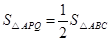 ����
����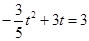 ��
��
��t=1�������淽�̲�������
�������һʱ��t��ʹ�߶�PQ��Rt��ACB���ܳ������ͬʱƽ�֣�
��4������P��PM��AC��M��PN��BC��N��
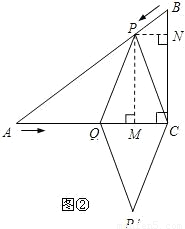
���ı���PQP'C�����Σ���ôPQ=PC��
��PM��AC��M��
��QM=CM��
��PN��BC��N����֪��PBN�ס�ABC��
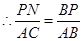 ��
��
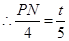 ��
��
��� ��
��
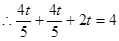
���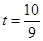 ��
��
 ��
��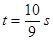 ʱ���ı���PQP'C�����Σ�
ʱ���ı���PQP'C�����Σ�
��ʱ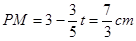 ��
��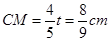 ��
��
��Rt��PMC��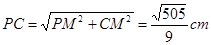 ��
��
������PQP��C�߳�Ϊ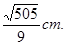
���㣺���⿼��������������ε��ۺ�Ӧ��
�����������Ĺؼ�����ȷ���������ߣ��ҵ����Ƶ������Σ�����������������εĶ�Ӧ�߳ɱ��������ʡ�