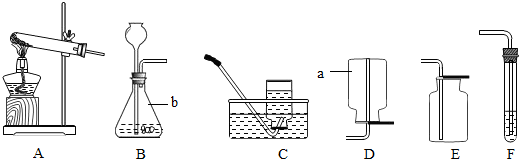
��Ŀ����
��ͼ��ʵ���ҳ��õ�ʵ��װ�ã���ش��й����⣺
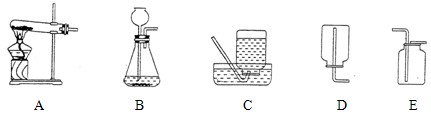
��1��װ��C�ɵ��ܡ�_______��_______�������������ƣ���ɡ�
��2��ʵ������H2O2��MnO2��ȡO2�Ļ�ѧ����ʽΪ__________________��ѡ�õķ���װ����_______������ţ���ͬ��������KClO3��MnO2��ȡO2ʱ����ѡ�õķ���װ����_________��
��3��ʵ������ȡCO2�Ļ�ѧ����ʽΪ__________________�����Ƶõ�CO2�л���HCl��H2O���ᴿʱ��NaOH�����ȥ���ʣ�����Ϊ���ַ���_______������С������С�����������______________��
��4��ʵ������ȡH2Ӧѡ�õ��ռ�װ����________��_________��
��2��ʵ������H2O2��MnO2��ȡO2�Ļ�ѧ����ʽΪ__________________��ѡ�õķ���װ����_______������ţ���ͬ��������KClO3��MnO2��ȡO2ʱ����ѡ�õķ���װ����_________��
��3��ʵ������ȡCO2�Ļ�ѧ����ʽΪ__________________�����Ƶõ�CO2�л���HCl��H2O���ᴿʱ��NaOH�����ȥ���ʣ�����Ϊ���ַ���_______������С������С�����������______________��
��4��ʵ������ȡH2Ӧѡ�õ��ռ�װ����________��_________��
(1)����ƿ��ˮ��
(2)2H2O2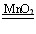 2H2O+O2����B��A
2H2O+O2����B��A
(3)CaCO3+2HCl��CaCl2+H2O+CO2���������У���ΪCO2Ҳ����NaOH��Ӧ
(4)C��D
(2)2H2O2
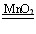 2H2O+O2����B��A
2H2O+O2����B��A(3)CaCO3+2HCl��CaCl2+H2O+CO2���������У���ΪCO2Ҳ����NaOH��Ӧ
(4)C��D

��ϰ��ϵ�д�
�����Ŀ