��Ŀ����
ij��ѧ�о���ѧϰС��Ϊ�ⶨ�г����۵�ʳ��������(���ҹ涨ʳ����������3.5 g/100 mL��5 g/100 mL)��������һ����������������������Һ��Ȼ����вⶨ����������С���ʵ���о������ش�������⣮
(1)����100 g��������Ϊ5��������������Һ��Ҫˮ________mL(ˮ���ܶ�Ϊ1 g/cm3)��
(2)��ʳ���е���������Ǵ��ᣬ�������������Ʒ�Ӧ�ķ���ʽΪ��
CH3COOH��NaOH��CH3COONa��H2O����ʳ��500 mL����ͬѧ����ȡ��10 mLʳ�ף�����5��������������Һ4.8 g��ǡ����ȫ��Ӧ����ͨ�������жϸ�ʳ���Ƿ�ϸ�(д���������)
�𰸣�

��ϰ��ϵ�д�
�����Ŀ

 ij��ѧ�о���ѧϰС����ѧϰ�ˡ����������������ⶨ��ʵ��Ļ����ϣ��Ľ��˽̲��е�ʵ�飬��Ƴ���ͼ��ʾ��ʵ��װ�ã�
ij��ѧ�о���ѧϰС����ѧϰ�ˡ����������������ⶨ��ʵ��Ļ����ϣ��Ľ��˽̲��е�ʵ�飬��Ƴ���ͼ��ʾ��ʵ��װ�ã�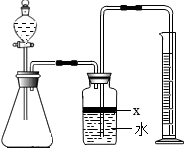 ��������AlN����һ�����������ϣ��㷺Ӧ���ڼ��ɵ�·��������ij�������к���̼�����������ʣ�ij��ѧ�о���ѧϰС����Ʋ����������ʵ�飮
��������AlN����һ�����������ϣ��㷺Ӧ���ڼ��ɵ�·��������ij�������к���̼�����������ʣ�ij��ѧ�о���ѧϰС����Ʋ����������ʵ�飮
