��Ŀ����
ij��ѧ��ȤС����ʵ��������200g��������Ϊ8%������������Һ��ʵ�鲽�����¡�
��1�����㣺��Ҫ�������ƹ��������Ϊ �� g��ˮ�����Ϊ �� mL��ˮ���ܶȽ��ƿ���1g/cm3����
��2������������������ƽƽ�⣬��һ���ձ�����������ƽ�� �� �̣�������������Ȼ�� �� �����������Ⱥ�˳��ѡ����ĸ����ֱ����ƽƽ�⡣
A�����������ƹ�������ձ��� B������Ҫ�������롢�ƶ�����
�ò��������ձ�������ֽ�����������Ƶ�ԭ���� �� ��
��3���ܽ⣺����Ͳ��ȡ�����ˮ������ʢ���������ƹ�����ձ�����裬ʹ���ܽ⣬����ȴ�����¡�
��4������õ���Һװ�� �� ���ϸ��ƿ�� ���ƿ���������� �� ���������������Ƥ�������������ϱ�ǩ��
��1��16 184 ��2���� BA ���������׳��⣨���������ƾ��и�ʴ�ԣ�
��4��ϸ��ƿ ��Ƥ��
���������������ƹ��������200g��8%=16g��ˮ������=200g-16g=184g��ˮ�����=184g��1g/mL=184mL��������������ʱ�������ң��������ƾ��и�ʴ�Բ���ֱ�ӷ���ֽ�ϳ�����Һ��ҩƷһ��װ��ϸ��ƿ�У����������ܺͲ����еĶ������跴Ӧ����˲����ò�������

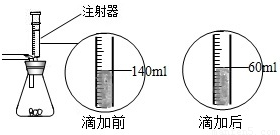
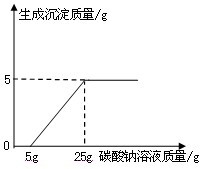 ij��ѧ��ȤС������ʯ��ʯ�����ʲ����ᷴӦ��Ҳ������ˮ����ϡ���ᷴӦ��ȡ������̼����������Ӧ��ķ�Һ������Һ��ʱ������ʵ��������һƿδ֪����������Na2CO3��Һ�����Ǿ������ø÷�Һ�ⶨ��ƿNa2CO3��Һ���������������������Ƚ���Һ���ˣ�Ȼ�����Һ�������μ�Na2CO3��Һ������Na2CO3��Һ�����������ɳ��������Ĺ�ϵ����ͼ��ʾ��
ij��ѧ��ȤС������ʯ��ʯ�����ʲ����ᷴӦ��Ҳ������ˮ����ϡ���ᷴӦ��ȡ������̼����������Ӧ��ķ�Һ������Һ��ʱ������ʵ��������һƿδ֪����������Na2CO3��Һ�����Ǿ������ø÷�Һ�ⶨ��ƿNa2CO3��Һ���������������������Ƚ���Һ���ˣ�Ȼ�����Һ�������μ�Na2CO3��Һ������Na2CO3��Һ�����������ɳ��������Ĺ�ϵ����ͼ��ʾ��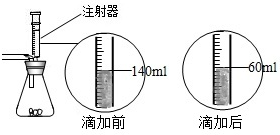 ij��ѧ��ȤС����ʵ�������ȡ������̼����ѡ������ͼ�ķ���װ�ã�����ȡ��Ϻ���ͬѧ�����װ�û��ܲ��ϡ���������������С��ͬѧ��ȡ12.5g��CaCO380%��ʯ��ʯ��ĩ��Ʒ����װ���У���ע�����μ�ϡ������ǡ����ȫ��Ӧ���μ�ǰ�����������ͼ��ʾ����������ϡ�����Ӧ��ϡ������ܶ�Ϊ1.25g/mL��
ij��ѧ��ȤС����ʵ�������ȡ������̼����ѡ������ͼ�ķ���װ�ã�����ȡ��Ϻ���ͬѧ�����װ�û��ܲ��ϡ���������������С��ͬѧ��ȡ12.5g��CaCO380%��ʯ��ʯ��ĩ��Ʒ����װ���У���ע�����μ�ϡ������ǡ����ȫ��Ӧ���μ�ǰ�����������ͼ��ʾ����������ϡ�����Ӧ��ϡ������ܶ�Ϊ1.25g/mL��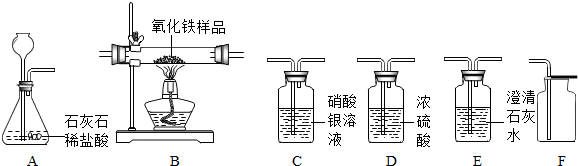
 ij��ѧ��ȤС����ʵ�������ȡ������̼����ѡ������ͼ�ķ���װ�ã�����ȡ��Ϻ���ͬѧ�����װ�û��ܲ��ϡ���������������С��ͬѧ��ȡ12.5g��CaCO380%��ʯ��ʯ��ĩ��Ʒ����װ���У���ע�����μ�ϡ������ǡ����ȫ��Ӧ���μ�ǰ�����������ͼ��ʾ����������ϡ�����Ӧ��ϡ������ܶ�Ϊ1.25g/mL��
ij��ѧ��ȤС����ʵ�������ȡ������̼����ѡ������ͼ�ķ���װ�ã�����ȡ��Ϻ���ͬѧ�����װ�û��ܲ��ϡ���������������С��ͬѧ��ȡ12.5g��CaCO380%��ʯ��ʯ��ĩ��Ʒ����װ���У���ע�����μ�ϡ������ǡ����ȫ��Ӧ���μ�ǰ�����������ͼ��ʾ����������ϡ�����Ӧ��ϡ������ܶ�Ϊ1.25g/mL��