��Ŀ����
ij�о���ѧϰС����������װ�ý����������ȡʵ�飬��ش��������⡣
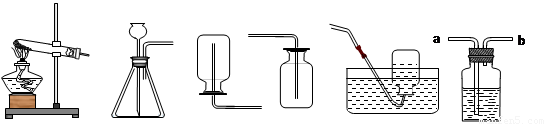

A B C D E F
��1��ѡ��Aװ����ȡ�����Ļ�ѧ����ʽ ����ȡ����ʱ����װ��������Ӧ�� ��������ҩƷ��ʹ��Fװ���ſ������ռ�����������Ӧ�� ��ͨ�루�a����b������
��2��A��E���ӣ���ȡ������������ԭ���� ��дһ�㼴�ɣ���
��3��ʵ������ȡ������̼ѡ��ķ���װ�ú��ռ�װ���� ����ѡ����ĸ����ѡ��÷���װ�õ������� �����������̼�Ƿ��ռ����ķ����� ��
��4������Fװ�ü����Ƶõ������Ƿ�Ϊ������̼����Fװ����Ӧ����������� ������Ӧ�� ��ͨ�루�a����b���������� ��
��1��2KMnO4 K2MnO4+MnO2+O2��
���װ�������� a ��2�����ܿڸ�������ð���Ϳ�ʼ�ռ�������ƿû��װ��ˮ�� ��3��BD �����Һ�巴Ӧ������Ҫ���� ��ȼ�ŵ�ľ�����ڼ���ƿ�ڣ���ľ��Ϩ���������̼���ռ����� ��4������ʯ��ˮ
a �����
K2MnO4+MnO2+O2��
���װ�������� a ��2�����ܿڸ�������ð���Ϳ�ʼ�ռ�������ƿû��װ��ˮ�� ��3��BD �����Һ�巴Ӧ������Ҫ���� ��ȼ�ŵ�ľ�����ڼ���ƿ�ڣ���ľ��Ϩ���������̼���ռ����� ��4������ʯ��ˮ
a �����
��������
�����������ΪAװ�����跴Ӧ�����Ǽ��ȣ����Թ�����һ�������ʣ�1��ѡ��Aװ����ȡ�����Ļ�ѧ����ʽ2KMnO4 K2MnO4+MnO2+O2������ȡ����ʱ����װ��������Ӧ�ȼ���װ�õ������ԣ�������ҩƷ��ʹ��Fװ���ſ������ռ���������Ϊ�������ܶȱȿ����Ĵ���������Ӧ��a��ͨ�룻��2��A��E���ӣ���ȡ������������ԭ���ǵ��ܿڸ�������ð���Ϳ�ʼ�ռ�����3��ʵ������ȡ������̼���ݷ�Ӧ�����ͷ�Ӧ���״̬ѡ��ķ���װ��Ϊ��Һ������װ�ú����ɵĶ�����̼�ܶȱȿ�������������ˮѡ���ռ�װ���������ſ����������Է���ΪBD����3��ʵ������ȡ������̼ѡ��ķ���װ�ú��ռ�װ���ǹ����Һ�巴Ӧ������Ҫ���ȣ����ݶ�����̼��ȼ��Ҳ��֧��ȼ�յ����ʣ����������̼�Ƿ��ռ����ķ����ǽ�ȼ�ŵ�ľ�����ڼ���ƿ�ڣ���ľ��Ϩ���������̼���ռ�������4������Fװ�ü����Ƶõ������Ƿ�Ϊ������̼����Fװ����Ӧ����������dz���ʯ��ˮ������Ӧ��a�˽�������Ϊ����ʯ��ˮ����ǣ�
K2MnO4+MnO2+O2������ȡ����ʱ����װ��������Ӧ�ȼ���װ�õ������ԣ�������ҩƷ��ʹ��Fװ���ſ������ռ���������Ϊ�������ܶȱȿ����Ĵ���������Ӧ��a��ͨ�룻��2��A��E���ӣ���ȡ������������ԭ���ǵ��ܿڸ�������ð���Ϳ�ʼ�ռ�����3��ʵ������ȡ������̼���ݷ�Ӧ�����ͷ�Ӧ���״̬ѡ��ķ���װ��Ϊ��Һ������װ�ú����ɵĶ�����̼�ܶȱȿ�������������ˮѡ���ռ�װ���������ſ����������Է���ΪBD����3��ʵ������ȡ������̼ѡ��ķ���װ�ú��ռ�װ���ǹ����Һ�巴Ӧ������Ҫ���ȣ����ݶ�����̼��ȼ��Ҳ��֧��ȼ�յ����ʣ����������̼�Ƿ��ռ����ķ����ǽ�ȼ�ŵ�ľ�����ڼ���ƿ�ڣ���ľ��Ϩ���������̼���ռ�������4������Fװ�ü����Ƶõ������Ƿ�Ϊ������̼����Fװ����Ӧ����������dz���ʯ��ˮ������Ӧ��a�˽�������Ϊ����ʯ��ˮ����ǣ�
���㣺ʵ������ȡ�����˼·�ͷ���

 �¿α�������������ҵ�������γ�����ϵ�д�
�¿α�������������ҵ�������γ�����ϵ�д�



