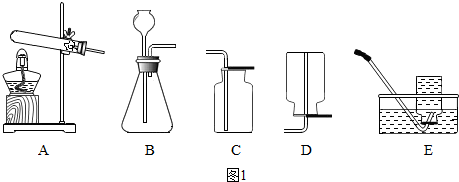
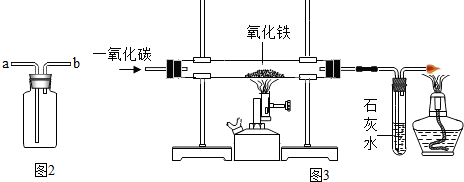
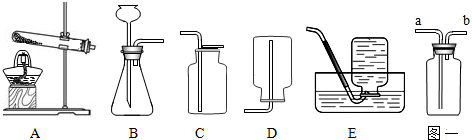
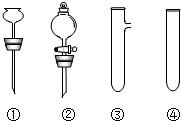
��Ŀ����
���������ʵ������ȡ���������������ʻش��������⣮
��1���������շ�������ȡ����������ȡ�����ԭ�Ͽ�ѡ��
a��ϡ������п�� b��ˮ��ͨ�磩 c��ϡ������ͭƬ d��ϡ����������
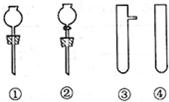
��2����Ҫ��װһ���Կ��������������ʵ�װ�ã�������ͼ��ѡ����ʵ�������
��3������������ԭ����ͭʱ����
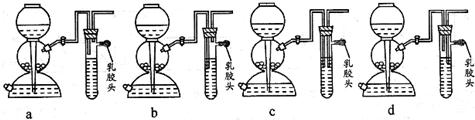
��4����ȼ�������������ܻᷢ����ը��Ϊ�˷�ֹ���⣬������һ����ȫװ�ã���ͼ��װ���������õ���
��1���������շ�������ȡ����������ȡ�����ԭ�Ͽ�ѡ��
a
a
��a��ϡ������п�� b��ˮ��ͨ�磩 c��ϡ������ͭƬ d��ϡ����������
��2����Ҫ��װһ���Կ��������������ʵ�װ�ã�������ͼ��ѡ����ʵ�������
�ڢ�
�ڢ�
�����ţ���
��3������������ԭ����ͭʱ����
ͨ����
ͨ����
���ټ��ȣ�ʵ�����ʱ����ֹͣ����
ֹͣ����
�����Թ���ȴ����ֹͣͨ��������4����ȼ�������������ܻᷢ����ը��Ϊ�˷�ֹ���⣬������һ����ȫװ�ã���ͼ��װ���������õ���
b
b
��
��������1��a������ʵ������������ԭ����ϡ�����ӷ����жϣ�
b��ˮ��ͨ�磩���������ĵ����жϣ�
c������ͭ�Ľ�������жϣ�
d������ϡ����Ļӷ����жϣ�
��2��Ҫ���Ʒ�Ӧ���ʣ��������ṩ���������ɲ��ÿ��Ƽ���Һ��ҩƷ���������ʵķ�������ѡ��Һ©����ͬʱѡ�ۣ���3�������Լ�ͭ�������ڼ��ȵ�����������������ͭ�ش�
��4����Ϊ��ȼ�������������ϵ�ȼ�ױ�ը����ν��ȫװ��Ӧ���ֿܷ����巢��װ�������װ�ã��Ҳ�Ӱ������ļ���ģ�
b��ˮ��ͨ�磩���������ĵ����жϣ�
c������ͭ�Ľ�������жϣ�
d������ϡ����Ļӷ����жϣ�
��2��Ҫ���Ʒ�Ӧ���ʣ��������ṩ���������ɲ��ÿ��Ƽ���Һ��ҩƷ���������ʵķ�������ѡ��Һ©����ͬʱѡ�ۣ���3�������Լ�ͭ�������ڼ��ȵ�����������������ͭ�ش�
��4����Ϊ��ȼ�������������ϵ�ȼ�ױ�ը����ν��ȫװ��Ӧ���ֿܷ����巢��װ�������װ�ã��Ҳ�Ӱ������ļ���ģ�
����⣺��1��a��ϡ������п���������Ȼ�п����������Ӧ�������У����������ռ�����ϡ����ȶ��ԣ��Ƶ�����ϴ���������ȷ��
b�����ˮҪ���Ĵ����ĵ��ܣ����Դ���
c��ϡ������ͭƬ����Ӧ���������������������Դ���
d��ϡ�����ܻӷ����Ȼ������壬�Ƶõ��������������Դ���
��2���������ṩ���������ɲ��ÿ��Ƽ���Һ��ҩƷ���������ʵķ�������ѡ��Һ©����ͬʱѡ������װ�âۣ�
��3����ȼ�������������Ϻ��ȼ������ױ�ը�����ȼ��Ⱥ�ͨ����������ʱ���ȵ�������������Ļ�����壬�з�����ը��Σ�գ�ʵ��������粻���Թ���ȴ���ֹͣͨ���������������Թܣ��������ȵ�ͭ��Ӧ�������ɺ�ɫ������ͭ��
��4��a�����巢��װ�������װ�ò��ֿܷ������Դ���
b����ȫװ��Ӧ���ֿܷ����巢��װ�������װ�ã��Ҳ�Ӱ������ĵ����ͼ��飬������ȷ��
c����װ�����������������Դ���
d����װ�����������������Դ���
�ʴ�Ϊ����1��a����2���ڢۣ���3��ͨ������ֹͣ���ȣ���4��b��
b�����ˮҪ���Ĵ����ĵ��ܣ����Դ���
c��ϡ������ͭƬ����Ӧ���������������������Դ���
d��ϡ�����ܻӷ����Ȼ������壬�Ƶõ��������������Դ���
��2���������ṩ���������ɲ��ÿ��Ƽ���Һ��ҩƷ���������ʵķ�������ѡ��Һ©����ͬʱѡ������װ�âۣ�
��3����ȼ�������������Ϻ��ȼ������ױ�ը�����ȼ��Ⱥ�ͨ����������ʱ���ȵ�������������Ļ�����壬�з�����ը��Σ�գ�ʵ��������粻���Թ���ȴ���ֹͣͨ���������������Թܣ��������ȵ�ͭ��Ӧ�������ɺ�ɫ������ͭ��
��4��a�����巢��װ�������װ�ò��ֿܷ������Դ���
b����ȫװ��Ӧ���ֿܷ����巢��װ�������װ�ã��Ҳ�Ӱ������ĵ����ͼ��飬������ȷ��
c����װ�����������������Դ���
d����װ�����������������Դ���
�ʴ�Ϊ����1��a����2���ڢۣ���3��ͨ������ֹͣ���ȣ���4��b��
�������������ȡ�dz�����Ҫ�Ļ�ѧʵ��֮һ���������ص㣬���ǿ����ȵ㣬���ⷴӦԭ��������װ�õ�ѡ�����ݡ�����ļ��顢�������ռ��������˽�ʵ�鲽���ע�������ǽ����������Ĺؼ���

��ϰ��ϵ�д�
 �ŵ������ϵ�д�
�ŵ������ϵ�д� 53������ϵ�д�
53������ϵ�д�
�����Ŀ