��Ŀ����
���ǵ��ճ������벻��������
��1���ؿ��к������Ľ���Ԫ����______��
��2�������ʯ��ʱ�����Ƚ�����ͭ��ʱ������д������̼��ԭ����ͭ�Ļ�ѧ����ʽ______��
��3�����й����ϱ�����ÿ������ʴ�����ϵĽ����൱���������20%��40%������M����¶���ڿ����У������ѱ���ʴ������⣬��ʴ���г�����M�⣬������C��H��O����Ԫ�أ����������غ㶨���Ʋ⣺ʹ����M��ʴ��������һ����______���ѧʽ����ͬ����������______��
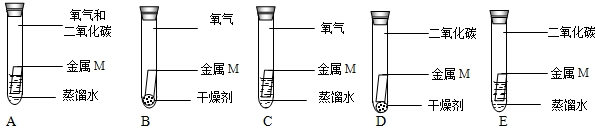
��4��С��Ϊ��֤�����е�O2�Ƿ�μ��˷�Ӧ���������ʵ�飬���б���Ҫ����ʵ����______������ţ���˵������������ˮ�����Ѹ����ȴ����������ֻ��ˮ�Ҳ����������ʷ�Ӧ��

��1���ؿ��к������Ľ���Ԫ����______��
��2�������ʯ��ʱ�����Ƚ�����ͭ��ʱ������д������̼��ԭ����ͭ�Ļ�ѧ����ʽ______��
��3�����й����ϱ�����ÿ������ʴ�����ϵĽ����൱���������20%��40%������M����¶���ڿ����У������ѱ���ʴ������⣬��ʴ���г�����M�⣬������C��H��O����Ԫ�أ����������غ㶨���Ʋ⣺ʹ����M��ʴ��������һ����______���ѧʽ����ͬ����������______��
��4��С��Ϊ��֤�����е�O2�Ƿ�μ��˷�Ӧ���������ʵ�飬���б���Ҫ����ʵ����______������ţ���˵������������ˮ�����Ѹ����ȴ����������ֻ��ˮ�Ҳ����������ʷ�Ӧ��

��1���ؿ���Ԫ�صĺ���˳��������������
�ʴ�Ϊ����������Al����
��2��̼��ԭ����ͭ����ͭ�Ͷ�����̼����ѧ��Ӧʽ�ǣ�2CuO+C
2Cu+CO2����
�ʴ�Ϊ��2CuO+C
2Cu+CO2����
��3����1��M�dz���¶���ڿ����еģ�����H��Դ��ˮ��C��Դ�ڶ�����̼���ɸ�����������ж�����Ҳ���ܲ��룬�ʴ�Ϊ��H2O��CO2��O2��
��3����Ϊ��������ˮ���Ǿ����Ѹ����ȴ�ģ�A��E��ʵ����̽�������������ֻ��A�л����⣬��A��E�Ƚ�˵��O2�μ��˷�Ӧ����ѡAE��
�ʴ�Ϊ����������Al����
��2��̼��ԭ����ͭ����ͭ�Ͷ�����̼����ѧ��Ӧʽ�ǣ�2CuO+C
| ||
�ʴ�Ϊ��2CuO+C
| ||
��3����1��M�dz���¶���ڿ����еģ�����H��Դ��ˮ��C��Դ�ڶ�����̼���ɸ�����������ж�����Ҳ���ܲ��룬�ʴ�Ϊ��H2O��CO2��O2��
��3����Ϊ��������ˮ���Ǿ����Ѹ����ȴ�ģ�A��E��ʵ����̽�������������ֻ��A�л����⣬��A��E�Ƚ�˵��O2�μ��˷�Ӧ����ѡAE��

��ϰ��ϵ�д�
 �㾦�½̲�ȫ�ܽ��ϵ�д�
�㾦�½̲�ȫ�ܽ��ϵ�д� Сѧ�̲���ȫ���ϵ�д�
Сѧ�̲���ȫ���ϵ�д�
�����Ŀ