��Ŀ����
������ˮ�����������������Ȼ��Դ��
��1�������к�������������______������ʱ�̶��벻������������Ϊ�����е�������______��
��2����Ȼˮ�к����������ʣ�ʵ���ҳ��ù��˵ķ�����ȥˮ�����������ʣ�������Ҫ�õ�����������Ʒ�У�����ţ�______��ijͬѧ�ڹ��˺�����Һ��Ȼ���ǣ����ܵ�ԭ����______��
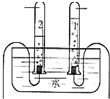
�ٴ���Ȧ������̨���ھƾ��ƣ���©���� �ܲ������� ����Ͳ�����ձ��� ����ֽ
��3��ij��ˮ��������ȡ��������С�ձ��У��������ˮ���裬�����ձ���û����ĭ��ȴ�д�����������˵���þ�ˮ��______���Ӳˮ������ˮ�������ճ�������ʹ��Ӳˮ����������鷳����ͥ�����г���������ˮӲ�ȵķ�����______��
��1�������к�������������______������ʱ�̶��벻������������Ϊ�����е�������______��
��2����Ȼˮ�к����������ʣ�ʵ���ҳ��ù��˵ķ�����ȥˮ�����������ʣ�������Ҫ�õ�����������Ʒ�У�����ţ�______��ijͬѧ�ڹ��˺�����Һ��Ȼ���ǣ����ܵ�ԭ����______��
�ٴ���Ȧ������̨���ھƾ��ƣ���©���� �ܲ������� ����Ͳ�����ձ��� ����ֽ
��3��ij��ˮ��������ȡ��������С�ձ��У��������ˮ���裬�����ձ���û����ĭ��ȴ�д�����������˵���þ�ˮ��______���Ӳˮ������ˮ�������ճ�������ʹ��Ӳˮ����������鷳����ͥ�����г���������ˮӲ�ȵķ�����______��
��1�������ijɷְ�������㣬��Լ�ǣ�����ռ78%������ռ21%���������������ǵ����������е������ܹ���������
��2������װ����Ҫ����Ȧ������̨��©�����ձ�������������ֽ����ֽ����Һ�������ֽ�ı�Ե��ʢ����Һ���ձ����ɾ��ȣ�����ɹ��˺���Һ��Ȼ���ǣ�
��3�������п��÷���ˮ������Ӳˮ����ˮ��������ĭ�϶ࡢ�������ٵ�����ˮ��������ĭ���١������϶����Ӳˮ���������⣬�øþ�ˮ��Ӳˮ����ͥ�����г�����еķ���������ˮ��Ӳ�ȣ�
�ʴ�Ϊ����1����������N2��������������
��2���٢ۢܢޢߣ���ֽ���𣨻�Һ�������ֽ�ı�Ե����ʢ����Һ���ձ����ɾ�����
��3��Ӳˮ����У�
��2������װ����Ҫ����Ȧ������̨��©�����ձ�������������ֽ����ֽ����Һ�������ֽ�ı�Ե��ʢ����Һ���ձ����ɾ��ȣ�����ɹ��˺���Һ��Ȼ���ǣ�
��3�������п��÷���ˮ������Ӳˮ����ˮ��������ĭ�϶ࡢ�������ٵ�����ˮ��������ĭ���١������϶����Ӳˮ���������⣬�øþ�ˮ��Ӳˮ����ͥ�����г�����еķ���������ˮ��Ӳ�ȣ�
�ʴ�Ϊ����1����������N2��������������
��2���٢ۢܢޢߣ���ֽ���𣨻�Һ�������ֽ�ı�Ե����ʢ����Һ���ձ����ɾ�����
��3��Ӳˮ����У�

��ϰ��ϵ�д�
�����Ŀ


 ������ˮ�����������������Ȼ��Դ����������˽���٣�
������ˮ�����������������Ȼ��Դ����������˽���٣�